Canada Gazette, Part I, Volume 156, Number 19: Biocides Regulations
May 7, 2022
Statutory authorities
Food and Drugs Act
Pest Control Products Act
Sponsoring department
Department of Health
REGULATORY IMPACT ANALYSIS STATEMENT
(This statement is not part of the Regulations.)
Executive summary
Issues: Products that sanitize or disinfect hard or soft non-living and non-liquid surfaces to prevent disease in humans or animals are collectively referred to as biocides. The need for health products, including biocides, has exponentially increased as a result of the COVID-19 pandemic. Certain biocides are able to kill SARS-CoV-2, the virus that causes COVID-19. These products are essential in a coordinated approach to help prevent the spread of COVID-19. To maintain a safe and effective supply of biocides for Canadians, and to alleviate certain pressures on the health care system and industry, interim orders and operational efficiencies were introduced to mitigate shortages of certain biocides in Canada. However, these are temporary measures that have been put in place to manage the response to the COVID-19 pandemic.
Biocides are currently regulated under separate regulatory frameworks in Canada with surface sanitizers and disinfectants having different associated requirements, despite having similar risks, benefits, uses, and ingredients. Having different requirements for these similar biocides has not provided a consistent approach to the regulation of these products, resulting in product classification challenges, differences in the administrative burden for the biocide supply chain, and potential hurdles for greater international alignment. These challenges were highlighted during the COVID-19 pandemic. The biocide industry has advocated for the implementation of a single regulatory framework for biocides that would address these challenges.
Health Canada is proposing to introduce modern regulations for biocides that would provide more flexibility in tailoring application and regulatory requirements specifically to these products to maintain a safe and effective supply of biocides for Canadians, and to facilitate future efforts in international alignment.
Description: Health Canada is proposing to create new regulations under the Food and Drugs Act (FDA) specific to biocides that would see
- the transfer of disinfectants that are currently regulated under the Food and Drug Regulations (FDR) and surface sanitizers regulated under the Pest Control Products Act (PCPA) that meet the definition of a drug to the proposed Biocides Regulations (the Regulations);
- the creation of a new, modern authorization and regulatory framework with safety, efficacy and quality regulatory requirements better suited to biocides;
- the maintenance of a life cycle approach to the regulation of biocides while supporting requirements and authorities for the continuous post-market monitoring of a biocide’s risks and benefits; and
- the creation of a pathway for authorization based on decisions from foreign regulatory authorities.
Rationale: The development of new regulations for the authorization and regulation of biocides, including a use of foreign decisions (UFD) pathway, would benefit Canadians and industry. Having harmonized regulations for both surface sanitizers and disinfectants as biocides that are modern and easy to understand would encourage industry to bring biocides to the Canadian market and would contribute to maintaining Canadians’ access to safe, effective, and high-quality products. This is critical to prepare for future emergency responses.
Regulating surface sanitizers as drugs under this new framework would subject manufacturers to regulatory requirements that are well suited to the nature of these products. Furthermore, the regulation of surface sanitizers under the FDA would harmonize regulatory requirements for all biocides, thereby reducing costs to industry and allowing for more timely authorization of these products.
The UFD pathway would enable Health Canada to leverage the authorization from foreign jurisdictions to bring biocides to the Canadian market sooner, while reducing duplication and the associated burden in the review of applications. This proposal would reduce costs to industry, and would encourage companies to bring their products to Canada without compromising on safety.
By harmonizing requirements, the proposed Biocides Regulations would also increase the predictability of the regulatory process for applicants. It would enhance the efficiency of Health Canada’s regulatory assessments through the consolidation of departmental scientific resources, and would support the biocide industry in bringing innovative products to market.
The total direct benefits of the proposed Biocides Regulations are estimated to be $61.6 million present value (PV) or $6.8 million annualized over a 15-year time period. This estimate is based on cost savings for both industry stakeholders and the Government of Canada from the reduced burden. The total direct costs of the proposal include a one-time cost to industry of $1.1 million PV for existing biocides to come into compliance with the proposed Biocides Regulations and $1.4 million PV over 15 years for the Government of Canada to update existing processes and implement the proposed Biocides Regulations. The result is a net benefit of $59 million PV or $6.5 million annualized over the next 15 years.
Issues
Developing tailored requirements through the creation of a single framework for biocides
The application and regulatory requirements set out in the Food and Drug Regulations (FDR) were developed to regulate pharmaceuticals and have not provided a consistent approach to the regulation of products that sanitize or disinfect hard or soft non-living and non-liquid surfaces (referred to as surface sanitizers and disinfectants in this document) to prevent disease in humans or animals. These products are collectively referred to as biocides. Biocides are distinct from pharmaceuticals in that they are intended to be applied to non-living surfaces only and are not authorized for the diagnosis or treatment of disease. This distinction also has an impact on Health Canada’s ability to identify, assess, and manage new risks associated with the use of these products. Due to these differences, the current application requirements and post-market safety measures for disinfectants set out in the FDR could be tailored to suit the risks and benefits associated with these products.
Hard and soft surface sanitizers are currently regulated under the Pest Control Products Act (PCPA) and have different requirements from disinfectants under the Food and Drugs Act (FDA). Furthermore, products with both disinfectant and sanitizer claims are regulated solely under the FDA.
Industry stakeholders have consistently identified a reduced administrative burden and opportunities for increased international alignment as priorities for regulatory modernization efforts. This includes the need for similar application requirements, which may be addressed through the implementation of a single regulatory framework that has requirements specific to biocides. Overall, Health Canada’s proposed Biocides Regulations would provide more flexibility in tailoring application and regulatory requirements specifically to biocides and would facilitate harmonization with international requirements.
Maintaining Canada’s supply of biocides
One of Health Canada’s roles is to regulate and authorize health products that improve and maintain the health and well-being of Canadians. The COVID-19 pandemic has created an unprecedented demand on Canada’s health care system. As a result of the pandemic, the need for health products, including disinfectants and surface sanitizers, has exponentially increased.footnote 1 In addition, Health Canada experienced challenges in responding to the volume of questions asked by biocide manufacturers during the pandemic regarding appropriate application pathways, application requirements and obligations. To alleviate certain pressures on industry and the health care system, interim orders and operational efficiencies were introduced to help increase the supply in Canada and mitigate shortages of certain products that sanitize or disinfect surfaces. However, these were only temporary measures that were put in place to manage the response to the COVID-19 pandemic. Unique regulations for biocides that include the ability to leverage a specific authorization in a foreign jurisdiction would remove barriers to both Health Canada and industry, providing Canadians with more timely access to biocides.
Many Canadian biocide suppliers have parent companies in other countries, predominantly in the United States (U.S.). The industry includes not only established large companies, but also small and medium-sized enterprises, bringing innovative products to global markets. In this context, Health Canada understands that international collaboration can be beneficial to promote the timely approval and import of a variety of safe and effective biocides. For the past several years, Health Canada has used information and data to support applications in other jurisdictions, when applicable, in order to better inform the review of biocides for market authorization in Canada. Health Canada is proposing to expand this practice to contribute to ensuring a Canadian supply of biocides is maintained that is safe, effective, and of high quality.
Background
Disinfectants and surface sanitizers are currently regulated under two different Acts. Disinfectants are regulated under the FDA and surface sanitizers are regulated under the PCPA. These products are assessed for their safety, efficacy and quality under different regulatory frameworks, with varying requirements, fees and timelines.footnote 2
Historically, disinfectants used in health care facilities, food processing establishments and on medical devices were regulated under the FDA, while institutional, agricultural, domestic and industrial uses of disinfectants were regulated under the PCPA. Products that could be used in either scenario (e.g. both household use and schools) had to be approved under both the FDA and the PCPA before they could be marketed.
In 2001, a regulatory amendment (PDF, 2.1 MB) exempted surface disinfectants that were used to prevent human and animal disease from the PCPA to consolidate the regulation of these products under the FDA. This amendment substantially reduced the duplicate reviews and approvals that were occurring under both the PCPA and the FDA. However, surface sanitizers continued to be regulated under the PCPA, with the exception of sanitizers used in food processing establishments; the latter continued to be regulated under the FDA to reduce safety concerns related to food products. Within this regulatory publication, Health Canada committed to re-examine transitioning surface sanitizer-only products from regulation under the PCPA to the FDA in the future. In addition, Health Canada recommended that products having both disinfectant and surface sanitizer uses be regulated solely under the FDA, because these products undergo a similar efficacy review and, by regulating them solely under the FDA, it would reduce duplicative reviews. As of April 2020, products with both surface disinfectant and sanitizer uses were transitioned from being regulated under the PCPA to the FDA.
Health Canada has mitigated shortages by implementing a number of measures and operational efficiencies in response to an increased demand for biocides during the COVID-19 pandemic. Health Canada permitted the temporary exceptional importation of disinfectants based on their authorization in foreign jurisdictions. This was done through the (ARCHIVED) Interim Order No. 2 Respecting Drugs, Medical Devices and Foods for a Special Dietary Purpose in Relation to COVID-19. Stakeholders have reacted positively to these measures overall, and approximately 280 disinfectants have been made available for exceptional importation and temporary sale in Canada.
In addition, Health Canada has experienced challenges responding to the significant increase in questions asked by biocide manufacturers during the pandemic regarding appropriate application pathways, application requirements and obligations. Due to the separate regulatory frameworks governing disinfectants and surface sanitizers, many of the inquiries Health Canada received during the pandemic pertained to questions on product classification. Overall, this situation has delayed the submission of applications and authorization of biocides, resulting in potential delayed access of these products to Canadians during the COVID-19 pandemic.
Health Canada is committed to supporting regulatory requirements that allow for a sufficient supply of biocides and to addressing some long-standing industry concerns. This proposal aligns with the intent of the Health and Biosciences Sectoral Regulatory Review Roadmap to identify and address regulatory barriers to economic growth and innovation. This proposal would improve Health Canada’s efficiency in approving a greater variety of biocides for the Canadian market without compromising safety, efficacy, and quality.
Objective
The objective of this proposal is to introduce a new regulatory framework under the FDA that is specific to the authorization and life cycle management of biocides. Tailored application and regulatory requirements for these products would facilitate greater international harmonization, reduce the administrative burden, provide stronger post-market oversight, and provide Canadians with more timely access to biocides.
Description
Biocides are currently regulated under separate regulatory frameworks in Canada with different requirements, despite having similar ingredients and associated risks and benefits. Health Canada is proposing new regulations under the FDA for biocides to address these differences that would see
- the transfer of disinfectants that are currently regulated under the FDR and surface sanitizers that meet the definition of a drug,footnote 3 for use on non-liquid surfaces regulated under the PCPA, to the proposed Biocides Regulations;
- the creation of a new, modern authorization and regulatory framework with safety, efficacy and quality regulatory requirements tailored to biocides;
- the maintenance of a life cycle approach to the regulation of biocides while supporting requirements and authorities for the continuous post-market oversight of a biocide’s risks and benefits; and
- the creation of a pathway for authorization based on decisions from other regulators, starting with the United States Environmental Protection Agency (U.S. EPA).
Scope
The scope of products that would be subject to the proposed Biocides Regulations would include disinfectants currently regulated under the FDR and surface sanitizers that meet the FDA definition of a drug. This includes disinfectants and sanitizers for use on hard or soft non-living and non-liquid surfaces. The following products would be excluded from the proposed Biocides Regulations:
- Sanitizers that do not meet the definition of a drug under the FDA. This includes algaecides, slimicides, material preservatives and products for odour control. These products would continue to be regulated under the PCPA;
- Sanitizers and disinfectants that meet the definition of a drug, but that are for use in air or water. This includes pool and spa disinfectants, water sanitizers, and air sanitizers. These higher-risk products would continue to be regulated under the PCPA;
- High-level disinfectant and sterilant solutions. This includes contact lens disinfectants and disinfectants intended for use on medical devices that are classified by Health Canada as invasive medical devices. These products would continue to be regulated under the Medical Devices Regulations;
- Disinfectants and surface sanitizers exclusively used directly on the surface of a food, such as fruits, vegetables and meat products. These products would continue to be regulated under the FDR by the Food Directorate of Health Canada as food processing aids;
- Drugs with antimicrobial activity for use on humans or animals, such as human use antiseptic drugs (e.g. hand sanitizers). These products would continue to be regulated under the FDR or the Natural Health Products Regulations; and
- Cleaners. These would continue to be regulated under the Canadian Consumer Product Safety Act (CCPSA) and the Hazardous Products Act.
Food-contact sanitizers and disinfectants are used for disinfecting and sanitizing surfaces that may come into contact with food in food premises, such as restaurants or processing plants. While food-contact disinfectants are regulated in the same manner as other disinfectants, the Pest Control Products Regulations (PCPR) exempt food-contact sanitizers used in commercial settings from the PCPA. Health Canada reviews these commercial food-contact sanitizers for acceptability for use in food processing establishments according to the requirements under the FDA. Food-contact sanitizers and disinfectants for use on surfaces in commercial or household settings would be included within the scope of the proposed Biocides Regulations.
Application and market authorization
Currently, the majority of disinfectants receive a drug identification number (DIN) under Division 1 in Part C of the FDR. Submissions for disinfectants with a new active ingredient or a new use receive a notice of compliance (NOC) under Division 8 in Part C of the FDR and a DIN. Surface sanitizers receive a pest control product registration under the PCPA.
The proposed Biocides Regulations would include an authorization pathway based on existing requirements in the FDR for disinfectants, but would be tailored uniquely to biocides. The authorization framework included in the proposed Biocides Regulations would be flexible enough to consider products with varying risks, benefits, and uncertainties, removing the need for different sets of requirements. The proposed Regulations would specify the application requirements and supporting information required to be eligible for a market authorization. To further support applicants, guidance would be made available by the date the proposed Biocides Regulations would be published in the Canada Gazette, Part II. The application would include requirements for the following information:
- General information: basic information on the product, applicant, manufacturer, and any importers to guide the type and level of review (e.g. brand name, directions for use, list of active ingredients and formulants).footnote 4
- Safety information: to help characterize the safety profile of the product (e.g. information concerning the toxicological/hazard profile of the product).
- Efficacy information: to help characterize the efficacy profile of the product (e.g. information demonstrating the product’s ability to meet its marketed claims).
- Quality information: to help characterize the identity, potency, and purity of the product.
Each biocide application that is received would undergo an assessment of the benefits and risks of the product before the Minister makes a decision on whether to issue a market authorization. Market authorization would be granted if the benefits of the product outweigh the risks, taking into consideration any associated uncertainties, provided that all information requirements are met. In addition, the Minister would be provided with the authority to request additional information outside of what was provided in the initial application, or to obtain a sample of a product for which an application for market authorization has been submitted (e.g. a sample of a product may be requested for product classification purposes) to determine whether the market authorization would be issued.
Applications based on comparison
The proposed Biocides Regulations would include an application pathway for biocides that rely on the comparison to a biocide that is already authorized in Canada. This application must include information demonstrating that
- the biocide has the same active ingredients in the same quantities as the other biocide;
- the formulants in the biocide are among those permitted to be included in the other biocide;
- the uses or purposes, intended users, settings and directions for use fall within the parameters of those approved for the other biocide;
- the risk information and directions for storage and disposal are the same as those approved for the other biocide; and
- the biocide has the same decontamination procedure as the other biocide, if the other biocide has such a procedure.
In addition, the applicant must attest that the biocide would be manufactured in accordance with the same master formula as the comparative biocide.
This pathway would reduce authorization costs and the length of time to make a regulatory decision since applicants would not have to provide all the information that is needed in a full review. Applicants using this pathway would not need to provide information related to the safety, efficacy and quality of the biocide, other than packaging information, as these requirements would be satisfied through the indication that the biocide is the same as the comparative biocide. Products receiving a market authorization through an application that is based on a comparative biocide would be required to follow the same post-market requirements that apply to all biocides under the proposed Biocides Regulations. The Minister would also be able to request any additional information relating to the biocide to make a determination on its benefits and risks.
Use of foreign decisions
The proposed Biocides Regulations introduce a use of foreign decisions (UFD) pathway that would allow biocide applicants to leverage the decision of a trusted foreign regulatory authority when applying for a market authorization for an identical product in Canada. This pathway would reduce authorization costs and the length of time to make a regulatory decision since applicants would not have to provide all the information that is needed in a full review. This would provide an incentive for companies to bring their product to the Canadian market, thus providing Canadians with access to a greater variety of products.
Health Canada would maintain a list incorporated by reference that identifies trusted foreign regulatory authorities whose regulatory decisions would be relied upon when reviewing a market authorization application. This list would be incorporated on an ambulatory basis. At this time, the list of trusted foreign regulatory authorities would be limited to the U.S. EPA. However, Health Canada would consider adding other trusted partners once a determination has been made that their standards for authorizing biocides for sale are able to meet the requirements in the Biocides Regulations and are consistent with Canada’s review processes.
Products approved through the UFD pathway would be required to follow the same post-market requirements that apply to all other biocides under the proposed Biocides Regulations. In the event that there is an issue that impacts the benefits, risks or uncertainties of the product approved through the UFD pathway, the Minister would be able to request the original data package that the market authorization holder (MAH) submitted to the foreign regulator. The Minister would also be able to request any additional information to make a determination on the benefits and risks of the product. The MAH would need to notify the Minister in the event that the authorization of the foreign biocide is recalled, revoked, or suspended. The MAH would also need to notify the Minister if the foreign regulatory authority required a product change to the foreign biocide.
Multiple products under one market authorization
Currently, each disinfectant authorized under Division 1 of the FDR requires a separate market authorization with a unique brand name for each formulation. Surface sanitizers regulated under the PCPA may have minor variations in formulants within one pest control product registration; however, each registration can only be associated with one brand name.
The proposed Biocides Regulations would allow for one application to be submitted for a market authorization that may contain multiple products with minor variations. The data package provided in the application would need to apply to all products outlined in the application, including any variations, and demonstrate the same risks and benefits associated with each product. Further, allowing multiple products with minor variations under one market authorization would decrease costs for biocide applicants and incentivize companies to bring their products to Canada.
This proposed application structure would align with those in place for biocides in the United States, the European Union, and the United Kingdom, and for surface sanitizers currently regulated under the PCPA. The U.S. EPA allows for minor variations in formulants to be registered under a single registration. Allowing for multiple products with minor variations in formulation within one market authorization would provide a consistent approach for applicants using the UFD pathway, as this aligns with the application structure in other international jurisdictions. The proposed Biocides Regulations would allow the following variations in products that stem from one application and market authorization:
- brand names;
- intended use or purpose;
- settings in which they are intended to be used;
- intended users;
- method of application;
- directions for use; and
- variations in formulants that are considered similar to one another.
All other aspects would need to be the same for all products stemming from one application and market authorization, including the following:
- active ingredients and associated quantities;
- physical form;
- storage information;
- shelf life;
- directions for decontamination;
- risk information;
- directions for disposal; and
- MAH name and contact information.
An application for multiple products under one market authorization would need to include additional information to support each variation in formulation, such as the following:
- confirmatory data, if the included formulants differ in other than fragrance or colour at concentrations of 1% or less of the product formulation to support the equivalence of the proposed variations in terms of safety, efficacy, and quality; and
- label text that reflects all proposed variations.
Figure 1: Visual representation of how multiple variations in a market authorization would translate into different products on the market.
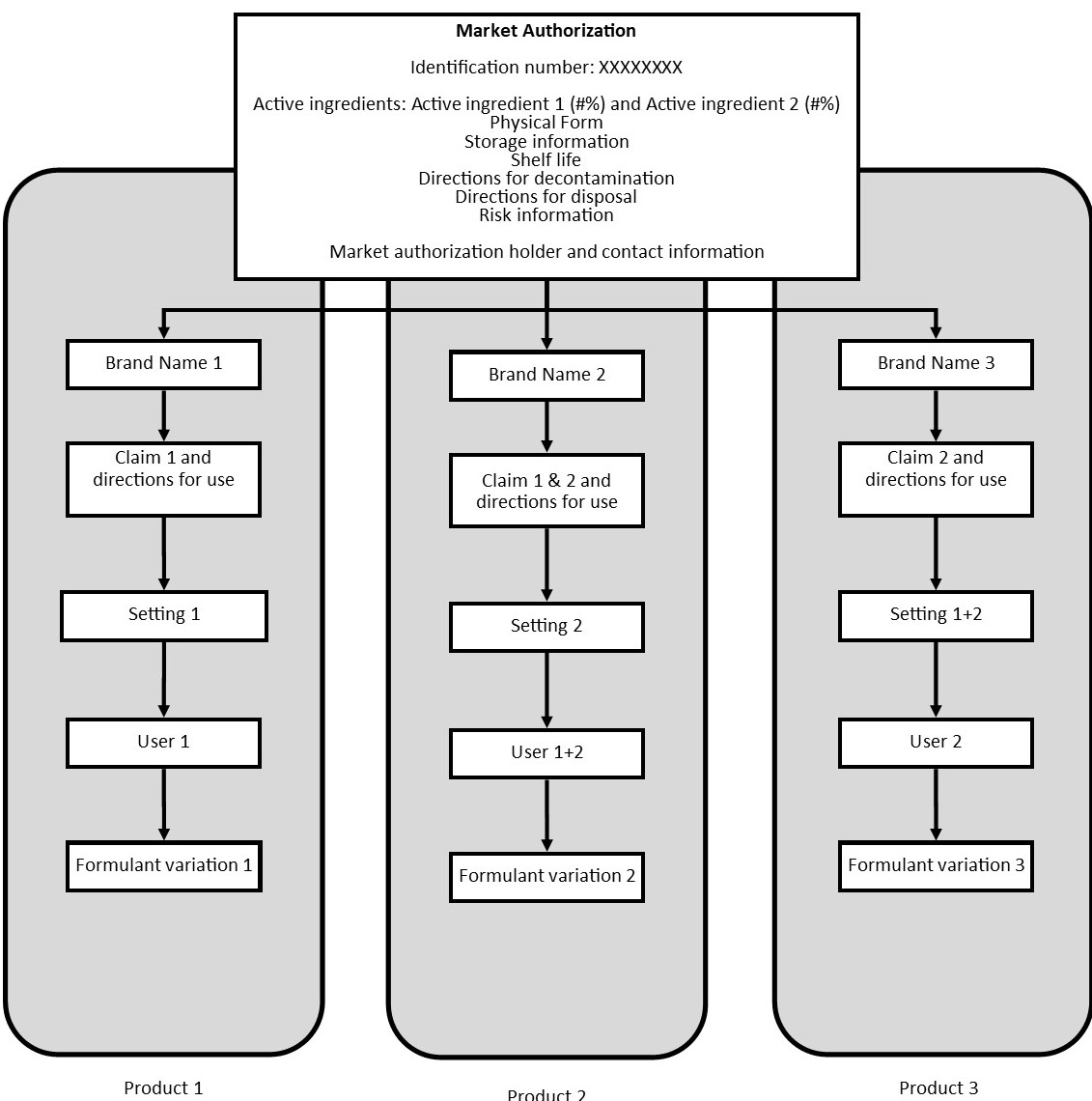
Figure 1 is visual representation of a market authorization that contains three different product variations. - Text version
Centered at the top of the figure there is a box containing the types of information in a market authorization that would be common across all variations of the product. This includes information such as:
- the identification number
- active ingredient 1 and active ingredient 2
- physical form
- storage information
- shelf life
- directions for decontamination
- directions for disposal risk information
- market authorization holder
- contact information
Three sets of arrows stem from the market authorization box outlining three different product variations a biocide approved under the market authorization could be marketed as.
Product 1 shows a product with:
- Brand Name 1
- Claim 1 and directions for use
- Setting 1
- User 1
- Formulant variation 1
Product 2 shows a product with:
- Brand Name 2
- Claim 1 and 2 and directions for use
- Setting 2
- User 1 and 2
- Formulant variation 2
Product 3 shows a product with:
- Brand Name 3
- Claim 2 and directions for use
- Settings 1 and 2
- User 2
- Formulant variation 3
Identification number
For disinfectants regulated under the FDR, each product is assigned an eight-digit drug identification number (DIN) that is unique to each drug product. The DIN is found on the product label and is searchable on the Drug Product Database (DPD), which is publicly available. Entries within the DPD outline key characteristics of drugs authorized by Health Canada. For surface sanitizers, a pest control product registration number is assigned to each product regulated under the PCPA. This registration number is searchable on the Pesticide Product Information Database, which allows the public to view information on products and active ingredients related to pesticides and other pest control products that are regulated by Health Canada.
For biocides, Health Canada is proposing to assign an eight-digit identification number. To minimize impact on existing disinfectant authorization holders, the eight-digit identification number would not need to change under the proposed Biocides Regulations. This identification number would be published as part of the biocide’s market authorization and included on the product label.
Amendments and notifications
Post-authorization product changes may have implications on the quality, risks, benefits and uncertainties of the biocide. In order for the Minister to assess the risk, benefits, and uncertainties associated with the product changes, the proposed Biocides Regulations would introduce the following categories of changes to a market authorization and associated actions to be taken by the product’s MAH:
Major changes
- Changes that could be reasonably expected to have a major impact on the quality, risks, benefits, or uncertainties associated with the biocide (e.g. a change to the directions for use or the addition of an intended use or purpose). The MAH would not be able to sell the changed product unless the MAH has submitted an application for a major change and the Minister has approved the change. Major changes would apply to all products under the market authorization.
Minor changes
- Changes that could be reasonably expected to have a minor impact on the quality, risks, benefits, and uncertainties associated with the biocide (e.g. addition of storage directions). The MAH would need to send a notification to the Minister outlining the change at least 30 days before making the change. If the change is not within the MAH’s control, the MAH would need to send a notification as soon as feasible after they become aware of the change.
- Changes to administrative information to the market authorization (e.g. a change in name or contact information of the MAH or an importer). The MAH would need to send a notification to the Minister as soon as feasible.
Changes that create a new biocide
- Changes that significantly alter the product would require the MAH to submit an application for a new market authorization. This would include any modifications to the active ingredients or a change to the physical form of the biocide.
Information about non-notifiable changes (i.e. changes that do not require an amendment or notification to the minister) would be outlined in guidance. This would include changes such as modifications to the format of the lot number on the label.
Packaging and labelling
Packaging
Currently, Health Canada receives limited information about the packaging of disinfectants (i.e. package size and container type), making it difficult to confirm that the packaging would maintain the product throughout its shelf life. Active ingredients in biocides may be corrosive in nature and can affect the integrity of the packaging of the product over time (e.g. may make it brittle). In addition, ingredients such as isopropyl alcohol and ethanol may evaporate over time. Appropriate packaging is necessary to maintain the stability and shelf life of a biocide product.
This proposal would require that packages must be able to contain the biocide under normal storage, display and distribution conditions (e.g. no leaks, can be reclosed) and must minimize product degradation and moisture loss. Packages would also need to be constructed to permit the withdrawal of the biocide in a manner that is safe to the user and the closure of the package to contain the biocide safely under normal conditions. These requirements are similar to what is required for surface sanitizers under the PCPR. In addition, the proposed Biocides Regulations would require applicants to specify the size and type of packaging used for the biocide and the properties and qualities of the packaging material. Health Canada intends to use this information to prevent the use of food- or beverage-like packaging for biocides. This would help to reduce the potential for accidental poisonings due to the biocide being mistaken for a food or beverage product. Similar restrictions on food- and beverage-like packaging are also imposed in the EU, the U.K., and Australia.
Labelling
The FDR prescribes specific information that disinfectant manufacturers are required to include on the principal display panel and other parts of the label. These requirements are tailored toward pharmaceutical labels, which tend to be much smaller in overall size and may come in additional packaging (e.g. cartons). Therefore, the labelling requirements currently outlined in the FDR would be tailored to biocides under the proposed Biocides Regulations.
The proposed Biocides Regulations would outline what information is required to be included on the principal display panel and other parts of the label and would require information to be clearly and prominently displayed in order to support the safe use of the product. Flexibilities would be provided so that manufacturers of products in small packages would be able to comply with the requirements. These requirements would align as much as possible with existing requirements in the FDR. This would help to accommodate the increase in demand for biocides as a result of the COVID-19 pandemic that are single use or of convenient size for consumers to use.
The proposed Biocides Regulations would also introduce a regulatory requirement for disinfectants to include the statement, “keep out of reach of children” to align with the current requirement for sanitizers.footnote 5 The proposed Regulations would also require surface sanitizers to include a biocide identification number (instead of the registration number of the pest control product), the expiry date,footnote 6 the physical form, and an indication that the product is sterile, if applicable, on the product label.
Post-market authorities and obligations
The post-market authorities proposed in the Biocides Regulations would enable Health Canada to support a life cycle approach to regulation by supporting continuous post-market oversight of a biocide’s risks and benefits. The proposed Biocides Regulations would carry over certain authorities available under the FDR related to safety reporting as well as Vanessa’s Law (e.g. the ability for the Minister to compel information, require tests and studies, impose terms and conditions, and conduct assessments after a market authorization has been issued). In addition, they would maintain Health Canada’s ability to identify, assess, and manage new risks and uncertainties associated with the use of biocides and to support compliance verification activities. The Minister would have the authority to direct a person to stop selling a biocide, or to suspend, in whole or in part, or to revoke a biocide’s market authorization, if necessary.
Notable incidents
The adverse drug reaction reporting requirements from the FDR would be carried over for biocides with some modifications.footnote 7 The term “adverse drug reaction” would be replaced by the term “notable incident,” which would be defined as any response to a biocide that adversely affects human health. A notable incident would also include any failures of effectiveness of a biocide that could have serious implications to human health (e.g. causes congenital malformations, chronic or significant disability, or is life-threatening). Notable incidents must be compiled by the MAH for the purposes of the safety monitoring requirements, described in more detail below.
Serious incidents
Currently for disinfectants, authorization holders must submit a report for serious adverse drug reactionsfootnote 8 that have occurred in Canada and serious unexpected adverse drug reactionsfootnote 9 that have occurred outside of Canada where there was a serious human health impact related to its use or exposure. Both of these reporting requirements would be carried over from the FDR for biocides with some changes. The terms “serious adverse drug reaction” and “serious unexpected adverse drug reaction” would be replaced with “serious incident” and “serious unexpected incident,” respectively.
A serious incident would be defined as a response to a biocide that results in serious implications to human health. A serious unexpected incident would be defined as a serious incident that is not identified in nature, severity or frequency in the risk information set out on the Canadian label of the biocide. The scope of serious incidents and serious unexpected incidents would be expanded to include failures of effectiveness that have caused serious implications to human health. The MAH would need to submit information about a serious incident or serious unexpected incident within 15 days after becoming aware of the information.
Hospital reporting
Mandatory reporting by hospitals would also apply to biocides for serious adverse drug reactions. These requirements would be carried over from the FDR and would require a hospital to report when exposure to the product results in serious harm to human health. Mandatory reporting would apply in situations when a serious adverse drug reaction has occurred, with the exception of failures of effectiveness that have caused serious implications to human health. The term “serious adverse drug reaction” is used to align with the language used in the FDA specific to hospital reporting. The hospital would be required to report the incident within 30 days after becoming aware of the information.
Safety monitoring requirements
Proposed safety and monitoring requirements have been adapted from the FDR, PCPA, Pest Control Products Incident Reporting Regulations (PCPIRR) and the CCPSA to better suit the unique traits of biocides. The new safety monitoring requirements would replace the existing annual summary report requirements under the FDR. The proposed Biocides Regulations would require the MAH to monitor the safety of their marketed biocides in a manner that allows them to detect safety issues that emerge because of real-world use. If a significant safety issue is detected, including a failure of effectiveness that may cause serious harm, the MAH would be required to notify Health Canada without delay. These activities, as well as information relating to incidents (described above), would need to be documented in a manner that is sufficient for auditing to verify compliance and support enforcement action, if necessary.
The MAH would be required to have procedures in place to compile and review both domestic and foreign information in a manner that ensures the effective and timely detection of safety issues regarding the biocide. This may include actions taken which relate to the biocide by foreign regulatory authorities (e.g. label changes, recalls, reassessments, risk communications, licence suspensions, and revocations) as well as failures in effectiveness of the biocide. These requirements would ensure that MAHs are self-monitoring their products to detect safety concerns.
Other post-market obligations
The proposed Biocides Regulations would carry over the following reporting and notification requirements that currently exist under the FDR for disinfectants:
- issue-related summary reports;
- notification of first sale;
- annual market notification; and
- notice of permanent discontinuance of sale.
Recalls
Recall system of control
The MAH and every person who sells a biocide, including importers, manufacturers, packagers, and labellers, would need to have a system of control in place that allows them to take effective and rapid action to recall a biocide from whom they sold it to, other than a consumer. Records that support the recall system would need to be retained and would include written procedures and sales records in a manner that enables traceability of the biocide. A recall may be necessary if the biocide presents a risk to the health of Canadians, or is in violation of the FDA or the proposed Biocides Regulations.
Recall reporting to Health Canada
The proposed Biocides Regulations would introduce recall reporting requirements for the MAH, importer, or manufacturer in Canada that are based on current guidance for the health product industry (including disinfectants) set out in Health Canada’s Drug and Natural Health Products Recall Guide (GUI-0039) and Recall Policy for Health Products (POL-0016).
The MAH, importer, or manufacturer would be required to notify the Minister within 24 hours of having made the decision to voluntarily recall a biocide from the Canadian market. The notification would include information to support Health Canada’s oversight of the recall, including
- details about the biocide being recalled, such as the brand name, identification number, affected lot numbers, date of manufacture, and expiry dates, if applicable;
- details about how and why the recall is being carried out, such as the reason for the recall, an assessment of the risk to health, a description of any other actions the MAH, importer, or manufacturer is taking; and
- details about the quantity of the biocide being recalled and information about its distribution within and outside of Canada.
The MAH, importer, or manufacturer would be required to submit the proposed communications to the Minister before starting the recall and after the recall has commenced, on request. Allowing Health Canada an opportunity to assess whether the communications are sufficient to mitigate the identified risk is an important step within the recall process.
Additional information would be provided to the Minister within 72 hours of having made the decision to recall, including
- the recall strategy, which would include information about how and when the Minister would be updated on the progress of the recall; and
- a description of the measures that the MAH, importer, or manufacturer intends to take to prevent a recurrence of the problem.
The MAH or importer would be required to provide the Minister with the results of the recall within 30 days of completing a recall, as well as more details about the corrective and preventive actions that have been, or will be taken.
Reporting requirements would also be introduced for any person who is ordered to recall a biocide through a ministerial order under section 21.3 of the FDA. The information to be provided would be consistent with requirements for voluntary recalls, with the exception of the time and manner, which would be specified by the Minister on a case-by-case basis.
Quality Control
The proposed Biocides Regulations would support the quality of biocides for sale in Canada through the inclusion of requirements based on current guidance for disinfectants set out in the Standard for the Fabrication, Control and Distribution of Antimicrobial Agents for Use on Environmental Surfaces and Certain Medical Devices (GUI-0049). These requirements include
- a prohibition on sale of a biocide unless it is manufactured, packaged, labelled and stored in accordance with the master formula;footnote 19
- requirements for manufacturers and packagers of sterile biocides; and
- a requirement for the MAH, importer, manufacturer, packager or labeller to investigate complaints that they receive with respect to the quality of the biocide and, if necessary, take corrective action.
Guidance would outline the expectation that MAHs establish quality agreements with any person they rely on for activities such as manufacturing, packaging and labelling. These quality agreements would allow the MAH to outline who is responsible for certain aspects of the quality of the biocide within the supply chain, facilitating the investigation of complaints and helping to ensure that appropriate corrective action is taken by the person responsible for the quality issue that is the source of the complaint. For example, a packager could be responsible for investigating complaints received with respect to the quality of the package.
Records
The MAH and any person within the supply chain that sells a biocide would be required to maintain records for biocides related to each activity that they conduct (manufacturing, packaging, labelling, as the case may be). Records would allow Health Canada to investigate product quality issues and provide assurance that the MAH, importer or a manufacturer is able to recall a product. The proposed Regulations would set out the following records that must be maintained:
- the master formula;
- evidence that each lot or batch of the biocide has been manufactured, packaged, labelled, and stored according to the master formula (i.e. an executed batch record);
- records to support the system of control for recalls, including records of sales containing sufficient information to enable the recall of every lot or batch of the biocide that has been made available for sale;
- a record of all complaints received in respect of the quality of the biocide, as well as any investigation and corrective action taken by the person responsible for the issue; and
- for biocides authorized by the UFD pathway, information used to support their foreign market authorization.
The retention period for quality control records would be one year past the end of the shelf life of the lot or batch of the biocide to which the record relates. If the shelf life is not known (e.g. in the case of a wholesaler), records would be retained for 6 years from the date the person sold the biocide. Records related to recalls are to be maintained until the expiry of the product. Records related to post-market safety monitoring and signal detection activities to support good vigilance practices would be retained by the MAH for 25 years, maintaining consistency with current practice for disinfectants and other drugs.
Other considerations
Data protection
Canada has international commitments, including under the Canada–United States–Mexico Agreement and the World Trade Organization Agreement on Trade-related Aspects of Intellectual Property Rights, to provide protection for agricultural chemicals containing new chemical entities from unfair commercial use of proprietary test data. In addition, Canada also has obligations under the Canada-European Union Comprehensive Economic and Trade Agreement to provide test data protection for plant protection products (i.e. pest control products). To meet these commitments, the PCPA and PCPR set out a data protection program for new active ingredients and their associated end-use products. Data protection is intended to both encourage the registration of new innovative pest control products and promote the availability of generic pest control products by outlining conditions that an applicant needs to follow when relying on an existing registrant’s data.
Registrants of some surface sanitizers registered under the PCPA have data protection in accordance with the PCPR. Unlike pharmaceutical drugs, disinfectants are not offered data protection under the FDR. To align with current practice for disinfectants under the FDR, the proposed Biocides Regulations would not offer data protection for biocides. For current surface sanitizer registrants under the PCPA, Health Canada proposes to allow them to maintain their registration under the PCPA for a period up to four years following the coming into force of the proposed Biocides Regulations.
Transition and coming into force
Coming into force
The proposed Biocides Regulations would come into force one year following the registration of the Regulations. This would provide industry with the necessary time to prepare their applications for new products according to the new requirements. Applicants that submit an application for a new market authorization would be required to meet all regulatory requirements in the Biocides Regulations following the coming-into-force date.
Transition
A phased transition strategy is proposed to allow existing registration and authorization holders of already-marketed surface sanitizers and disinfectants to clear current stock, update labels (if required) within their normal business cycle, and update internal processes to comply with post-market requirements. This is expected to reduce environmental impacts and compliance costs associated with meeting the proposed requirements.
Existing authorization holders of disinfectants and registrants of surface sanitizers who wish to continue to market their product as a biocide would be required to file an abbreviated application and receive a market authorization under the proposed Biocides Regulations within four years following the coming-into-force date of the proposed Biocides Regulations. They would be exempt from providing information relating to the benefits and risks of the biocide, as well as the specifications of the biocide, provided there are no changes being proposed to the product. In addition, existing authorization holders of disinfectants would be exempt from providing label text. Authorization and registration holders that fail to transition their products within the transition period would be considered out of compliance with the FDA and the proposed Biocides Regulations, and may be subject to compliance and enforcement action. These registration and authorization holders would be required to submit a full product application for review to receive a market authorization under the proposed Biocides Regulations.
As surface sanitizer registrations under the PCPA need to be renewed every five years, registrants may file an abbreviated application under the proposed Biocides Regulations within the four-year transition period, as their product registrations are set to expire. Registrants of surface sanitizers with both sanitizer and pesticide-related claims on their product would also have the option of maintaining their registration under the PCPA and removing all information relating to their biocide uses from their registration and product labels. Registrants of surface sanitizers with only biocide claims would only have the option to transfer over to the proposed Biocides Regulations.
Regulatory development
Consultation
Health Canada held an initial discussion with three industry associations in early July 2019: the Canadian Consumer Specialty Products Association (CCSPA), the Association pour le développement et l’innovation en chimie au Québec (ADICQ) and the Groupement provincial de l’industrie du médicament (GPIM). These associations provided feedback during this initial discussion and subsequently provided Health Canada with written comments.
During these initial consultations, industry voiced support for including surface sanitizer regulation under the FDA and the implementation of a UFD pathway. These measures are anticipated to support businesses to stay competitive globally and to provide additional access to safe and effective biocides for Canadians. Some raised concerns, however, that the UFD pathway would be disadvantageous for companies that do not sell biocides in the United States. This is because there is no reciprocal process for Canadian manufacturers to enter the U.S. market. From this perspective, Health Canada has analyzed the impacts of this proposal on small businesses and has included this analysis in the “Small Business Lens” section.
Throughout the COVID-19 pandemic, Health Canada has closely collaborated with industry stakeholders to help increase the availability of biocides to Canadians. Regularly scheduled meetings were held with key stakeholders where issues related to shortages and challenges surrounding application processes and requirements were discussed. Health Canada has taken the lessons learned from this pandemic to inform policy for the development of the proposed Biocides Regulations.
Stakeholder consultations on the proposed Biocides Regulations took place on April 7, 15, and 20, 2021. Stakeholders that attended the sessions included representatives from three major industry associations, namely the CCSPA, Food, Health and Consumer Products of Canada (FHCP), and the GPIM. Key components of the framework were discussed at the sessions, including scope, market authorization, packaging and labelling, post-market powers and obligations, and stakeholders were provided the opportunity to submit additional written feedback following these sessions.
Stakeholder feedback
Overall, stakeholders expressed their strong support for the proposed Biocides Regulations during these stakeholder sessions, as the regulation of similar products under different regulatory schemes has been a long-standing industry irritant.
Stakeholders voiced support for certain application provisions and labelling requirements, the removal of the annual summary report requirement, continued exemptions from establishment licensing and good manufacturing practices requirements, and the introduction of a UFD pathway. Some stakeholders advocated for the UFD pathway to be expanded to include more trusted regulatory authorities at a future date.
Concerns that were raised during these sessions included the scope of products that would be subject to the proposed Biocides Regulations, certain post-market requirements, including the retention period of some types of records, the threshold for incident reporting, the inclusion of a prohibition on food- and beverage-like packaging, the introduction of certain transparency measures, and the lack of provisions around the distribution of biocides as samples. Some stakeholders expressed the desire to have an application framework that would allow an applicant to obtain one market authorization that could include multiple products with minor variations in brand names, formulants, and uses or purposes; this would also simplify the process for filing changes with Health Canada. In addition, stakeholders expressed that a seven-year transition period would be preferable to limit costs.
Departmental response
During these sessions, while stakeholders were supportive of limiting the scope of foreign regulatory authorities to the U.S. EPA for the immediate future, they did express their desire to expand the List of Foreign Regulatory Authorities in the future to include Pharmaceutical Inspection Co-operation Scheme (PIC/S) countries and countries involved in mutual recognition agreements (MRA) with Canada. Health Canada would consider adding other foreign regulatory authorities once there is an understanding of their review processes and standards for authorizing biocides for sale in their respective jurisdictions.
Certain industry stakeholders advocated for the inclusion of air sanitizers in the proposed Biocides Regulations and the exclusion of food-contact sanitizers. Health Canada intends to regulate food-contact sanitizers under the proposed Biocides Regulations to provide a single regulatory framework for similar products. Air sanitizers have different health risk considerations regarding routes of exposure (i.e. inhalation or dermal exposure) and have not been included within the scope of biocides due to their high-risk nature. This decision could be revisited in the future should these higher-risk products be considered appropriate to include under the proposed Biocides Regulations.
There were some concerns raised with respect to certain proposed post-market requirements related to record retention and incident reporting. Industry advocated for a six-year record retention period for recall purposes to align with the record retention periods outlined for consumer products, such as cleaners. However, Health Canada is proposing to require manufacturers to maintain records to facilitate a recall for at least one year past the product’s shelf life. This would help to ensure that Health Canada is able to access records for products that have a shelf life of longer than five years. Regarding incident reporting, industry stakeholders indicated they receive product complaints from consumers that do not impact safety or efficacy of the product. Investigation of complaints related to the quality of the biocide is an important element of quality control and would be a regulatory requirement. Health Canada clarified that incident reporting should only capture credible incidents, as determined by some investigation by the MAH and should not capture unsupported complaints.
During the COVID-19 pandemic, Health Canada experienced issues with hand sanitizers packaged in beverage-like containers that increased the risk of unintentional ingestion. Industry raised concerns with respect to a prohibition on the use of food- and beverage-like packages for biocides, because it was not viewed as a risk in relation to these products. However, ingestion of biocides could cause serious harm, particularly in children. Health Canada is not proposing a specific prohibition on the use of food- and beverage-like packaging in the proposed Biocides Regulations. However, Health Canada is proposing to include application requirements that relate to the packaging of the biocide to prevent the use of food- and beverage-like packaging through a pre-market review. In addition, Health Canada would continue to enforce provisions outlined in the FDA that prevent a person from packaging any drug in a manner that is false, misleading, or likely to create an inaccurate impression of its risks and benefits.
Industry raised concerns with the measures Health Canada is taking to publish product information and information with respect to Health Canada’s decisions on product authorizations. They implied that this transparency might negatively affect their competitiveness in the market. Health Canada is committed to providing transparency on its decisions for product authorizations so that Canadians can make informed choices. Health Canada would publish the names of all ingredients, including formulants, in the biocide and the quantities of active ingredients as part of the market authorization. However, companies would not be required to include formulants on their product labels. The proposed Biocides Regulations would clarify that information that is published as part of the market authorization would cease to be confidential information when the market authorization is issued.
Regarding industry’s desire to include an ability for companies to engage in direct-to-consumer sampling, Health Canada would examine the appropriateness of the suggestion in future regulatory packages.
The proposal that was presented for consultation included a market authorization structure that only allowed for one product or brand name per authorization, which raised concerns from industry stakeholders. They had communicated that the U.S. EPA allows for multiple similar products per authorization and that allowing a similar market authorization in Canada would decrease burden by decreasing the number of overall applications required for authorization and amendments. Health Canada has addressed these concerns by proposing to allow multiple similar products per authorization.
The transition period for the proposed Biocides Regulations would be similar to that of other regulatory proposals that impact product labels. Upon registration of the proposed Regulations, existing authorization or registration holders would have up to five years to bring their products into compliance with these new requirements. This should provide sufficient time for companies to incorporate necessary changes within their regular business activities and minimize costs to the extent possible.
A costing survey for the Cost-Benefit Analysis was introduced at stakeholder sessions conducted in April 2021. The survey was formally distributed by email to stakeholders for their response by June 25, 2021.
Instrument choice
Health Canada examined a range of regulatory options before proposing separate regulations for biocides. Each option was assessed using the objectives of reducing the burden on Health Canada and the biocide industry while maintaining timely access of biocides for Canadians.
1. Transfer the regulation of surface sanitizers to the FDR from the PCPA and introduce regulatory requirements specific to biocides and a UFD pathway through the proposed Agile Regulations for Licensing Drugs
The intent of the proposed Agile Regulations for Licensing Drugs (Agile Framework) is to implement an agile, modern licensing scheme for drugs, which include disinfectants. The proposed Agile Framework would be implemented through a phased approach, and would introduce regulatory tools to manage risks and uncertainties (e.g. terms and conditions, risk management plans), introduce more targeted enforcement powers (e.g. suspension and revocation), and create formal transparency requirements to disclose information about the risks, benefits and uncertainties of drugs. In addition, the proposed Agile Framework would provide more flexible requirements and additional post-market authorities for biocides than are included in the FDR. This would allow for more efficient regulation of innovative products and would better manage the risks and uncertainties that are associated with a UFD pathway.
Phase I of the Agile Framework was included in the Forward Regulatory Plan 2021-2023 of the Department of Health (the Department). The introduction of a UFD pathway in the Agile Framework is planned for a future phase.
This option was not pursued, as the delay of introducing more tailored requirements for biocides and a UFD pathway until the Agile Framework comes into force may unnecessarily delay the expected benefits of this proposal to Canadians, industry and Health Canada. The increase in demand for biocides by Canadians and the number of submissions received by Health Canada during the COVID-19 pandemic has highlighted the urgent need for a more agile licensing and regulatory framework that is specific to biocides.
In addition, a regulatory framework for biocides that includes pharmaceutical products may unnecessarily hinder international harmonization and alignment for biocides, as any proposed changes would need to be considered in the context of a multitude of significantly different products (e.g. pharmaceuticals, biologics, vaccines and radiopharmaceuticals).
2. Transfer the regulation of surface sanitizers to the FDR from the PCPA and introduce a UFD pathway for biocides in the FDR
This option would allow for the regulation of both disinfectants and surface sanitizers under the FDR and would provide a UFD pathway for biocides, in advance of the proposed Agile Framework. As the regulatory amendments to the FDR would be minimal, this option would have the earliest coming-into-force date when compared with other options.
The regulation of both surface sanitizers and disinfectants (as biocides) under the same regulations would reduce stakeholder confusion and subject all biocides to similar requirements. Separate sets of regulations for similar products have caused confusion, as companies seeking authorization may not clearly know whether the FDR or PCPA application requirements apply to their products. This confusion can delay the entry of products into the Canadian market as they may need to set up pre-submission meetings or conduct testing before filing under the appropriate framework.
However, this option was not pursued for several reasons. Existing regulatory requirements in the FDR are tailored to pharmaceutical products. Many required protocols and tests to demonstrate safety and efficacy in humans (such as clinical studies) are not applicable to biocides. Even within the FDR, disinfectants are currently subject to different requirements depending on if they are authorized through Part C, Division 1 or Division 8. Introducing a UFD pathway into the FDR for biocides would have been challenging as the application requirements for disinfectants within the FDR differ from those internationally, making it difficult to ensure similar authorization thresholds. In addition, the FDR also contains fewer post-market authorities, relative to what was considered for inclusion in the proposed Agile Framework. These more robust post-market authorities are needed to effectively manage the risks and uncertainties associated with regulating innovative products or introducing a UFD pathway.
3. Create a new regulatory framework with requirements specific to biocides, outside of the FDR, that includes a UFD pathway (the proposed Biocides Regulations)
The proposed Biocides Regulations would allow for the authorization and regulation of disinfectants and surface sanitizers under a single framework, and separately from other health products under the FDA and pest control products under the PCPA. Separate sets of regulations for similar products have caused confusion, as companies seeking authorization may not clearly know whether the FDR (Part C, Division 1 or Division 8) or PCPA application requirements apply to their products. Subjecting these similar products to the same requirements would reduce confusion and increase the predictability of the regulatory process for applicants, enhance the efficiency of Health Canada’s regulatory assessments through the consolidation of scientific resources, and would support the biocide industry in reducing the administrative burden. These efficiencies would provide Canadians with more timely access to biocides.
In addition, the proposed Biocides Regulations would include post-market authorities that would support a flexible approach and more effectively manage the regulation of biocides with varying risks, benefits and uncertainties that may come with the regulation of innovative products and a UFD pathway.
The separation of the proposed Biocides Regulations from the proposed Agile Framework would allow these proposed Regulations to come into force sooner, expediting the expected benefits to Canadians, industry, and Health Canada. In addition, the proposed Biocides Regulations would address the concern that a regulatory framework for biocides that includes pharmaceutical products may unnecessarily hinder international harmonization and alignment.
It is for these reasons that Health Canada decided to proceed with the proposed Biocides Regulations as the preferred option.
Regulatory analysis
Benefits and costs
The cost-benefit analysis (CBA) aims to quantify the benefits and costs of the proposed Biocides Regulations. The new regulations proposed under the FDA for biocides aims to reduce the burden on industry and the Government by more closely aligning international and domestic requirements. This framework would see the transfer of disinfectants that are currently regulated under the FDR and surface sanitizers regulated under the PCPA to the proposed Biocides Regulations under the FDA, so that these similar products can be subject to the same regulatory requirements. A costing survey was shared in April 2021 with industry stakeholders for their input to assist with data collection for this CBA. All calculations for the costs and benefits are projected over a 15-year period, and the net present value is discounted by 7% as required by the Treasury Board Secretariat of Canada. The methodology underlying the analysis is available upon request.
Baseline scenario versus regulatory scenario
In the baseline scenario, surface sanitizers would continue to be regulated under the PCPA with the exception of sanitizers used in food processing establishments, which are regulated under the FDA. In addition, surface sanitizer registrations would be associated with only one brand name. In the regulatory scenario, surface sanitizers would be regulated as biocides under the FDA separately from other health products under the FDA and pest control products under the PCPA. Under the proposed Biocides Regulations, surface sanitizer applicants would be able to submit one application for a market authorization that may contain multiple products with minor variations.
In the baseline scenario, disinfectants that are used to prevent human and animal disease would continue to be regulated under the FDA. Each disinfectant authorized under Division 1 or Division 8 of the FDR would require a separate market authorization with a unique brand name for each formulation. Products with both surface disinfectant and sanitizer uses would be solely regulated under the FDA. In the regulatory scenario, disinfectants would be regulated as biocides separately from other health products under the FDA. Under the proposed Biocides Regulations, disinfectant applicants would be able to submit one application for market authorization that may contain multiple products with minor variations.
In the baseline scenario, for both disinfectants under the FDA and surface sanitizers under the PCPA, the UFD pathway does not exist. Under the regulatory scenario, in addition to creating regulations for similar products under the FDA, the proposed Biocides Regulations would introduce a UFD pathway. This UFD pathway would reduce application processing costs by allowing Health Canada to rely on information that a trusted foreign regulator has already reviewed and approved (e.g. information regarding the benefits, risks and uncertainties of the product). This pathway would also reduce authorization costs for industry and the length of time to receive a regulatory decision, thereby providing an incentive for businesses to bring their product to market. Overall, this would contribute to ensuring a Canadian supply of biocides is maintained that is safe, effective, and of high quality. Key assumptions are as follows:footnote 20
- All costs and benefits are presented in 2021 dollars;
- A discount rate of 7% was used in the analysis;
- The proposed Regulations would provide industry with a four-year transition period following a one-year coming-into-force period;
- The analysis evaluates the costs and benefits over a 15-year period; thus, the study period is presented from 2023 to 2037;
- The regulatory proposal would replace the existing process for surface sanitizers under the PCPA and disinfectants under the FDR to permit the sale of biocides on the Canadian market;
- Based on Health Canada’s internal database, this analysis assumes, within the baseline, an average annual growth rate for biocides of 8%. This aligns with the 7.7% growth rate projected in the United States;footnote 12,footnote 13
- As of 2021, 915 domestic and 335 international companies were involved in manufacturing biocides in Canada. It is unclear which of these international companies did not have some level of business conducted within Canada; therefore for the purposes of assessing the costs and benefits, this analysis assumes that all companies are in scope. However, for the UFD pathway, this analysis assumes that only 70% of companies would benefit from application cost savings.
- As of June 2021, there were 69 surface sanitizers currently on the market regulated under the PCPA that would be transitioning to the FDA and 1 919footnote 14 disinfectants that were currently on the market regulated under the FDA; and
- Manufacturers of biocides would most likely opt to wait until the last year of the transition period to file an abbreviated application under the proposed Biocides Regulations and update labels (if required) to exhaust their current stock of labels and products. This would allow them to reduce product and label waste and have more time to design their new labels.
Costs
The total direct cost to both industry and to the Government of Canada is estimated to be $2.5 million present value (PV) or $279,033 annualized over a 15-year period.
Costs to industry
The total direct cost to industry for the proposed Biocides Regulations includes the one-time cost of updating some labels, an application cost to change labels for products regulated under the PCPA and the one-time cost for existing registrants and authorization holders to submit an abbreviated application.footnote 15 These are anticipated to result in a total one-time cost of $1.6 million in year 5, the final year of the transition period, or $1.1 million PV over a 15-year period.
Label change costs
The FDR and PCPR currently prescribe specific information that is required to be included on the principal display panel and other parts of the label for disinfectants and surface sanitizers, respectively. For example, disinfectant authorization holders are required to include the lot number, expiry date and physical form on the inner and outer labels, whereas surface sanitizer registrants, for example, are required to include the settings for use and procedure for decontamination on the inner and outer labels. In addition, they are required to include a pest control registration number, a statement to read the label before use, and a statement to keep out of reach of children on the principal display panel.
The labelling requirements for biocides under the proposed Biocides Regulations would be aligned as much as possible to the current requirements for disinfectants under the FDR. Where relevant, certain requirements that currently exist for surface sanitizers under the PCPR have been included in the proposed Biocides Regulations. For biocides, a “DIN” designation and identification number would be required on the label. Currently for disinfectants under the FDR, the DIN is a requirement on the label. For surface sanitizers, the pest control registration number would be replaced with a “DIN” designation and an identification number as a new requirement. The proposed Biocides Regulations would also introduce a requirement for disinfectants to include the statement, “keep out of reach of children”. However, based on industry responses to the CBA survey and stakeholder consultations, it is assumed that disinfectants currently display this statement as recommended in guidance, and therefore would not be considered an incremental cost.
The proposed Biocides Regulations would require surface sanitizers to include an identification number with a “DIN” designation, the expiry date, and the physical form on the product label. These labelling requirements are already recommended in guidance and would not be considered new to the majority of industry stakeholders, with the exception of introducing the DIN for surface sanitizers. Based on industry responses to the CBA survey, as well as internal government data, it is assumed that the majority of industry already complies with the rest of the proposed labelling requirements. There are currently 69 surface sanitizers on the market for which the labels would be expected to be updated to incorporate the new biocide DIN labelling requirements. It is assumed that the cost to conduct a label change to incorporate these differences is $7,608 per product. Industry provided an average cost estimate in the CBA survey that ranged from $7,608 to $25,942 per stock keeping unit (SKU). For the purposes of the CBA, Health Canada assumes that the lower-bound range of the cost estimate best represents a proxy for addressing these minor labelling differences and the type of label printing process used (new cylinder heads, new steel plates, etc.). The lower-bound range is a suitable proxy as industry is provided with a long transition period that falls within the label redesign life cycle of five years. The lower-bound estimate was further validated with industry during consultations.
In addition, it is assumed that for half of the 69 surface sanitizers the registrants would choose to continue their registrations under the PCPA for uses that are specific to pest control products. As a result, consistent with the Pest Control Products Fees and Charges Regulations, these surface sanitizer registrants will be required to make label amendments under the PCPA at a cost of $1,204 per application per product to remove biocide uses that would be subject to the FDA.footnote 16 This is in addition to the cost of the label change.
These labelling requirements for surface sanitizers would result in a one-time cost of $566,490 at year five, the final year of the transition period.footnote 17
Reapplication costs
An abbreviated application would need to be filed for existing disinfectants and surface sanitizers under the proposed Biocides Regulations within four years following the coming-into-force date in order to obtain an authorization. An abbreviated application for surface sanitizers would include their product’s current and proposed label text, an attestation form, and an application form outlining company contact information and information about the product such as its brand name, physical form, active ingredient(s) and formulant(s), conditions of use, and packaging information. An abbreviated application for disinfectants would include the same attestation and application form as the surface sanitizer abbreviated application but would not include label text. This would be considered a one-time cost to registrants or authorization holders of existing products.
Based on previous cost estimates outlined in the Regulations Amending the Food and Drug Regulations (Human Milk Fortifiers), the cost to submit a product’s label text and an attestation could cost approximately $12,000 per product, which is used as a proxy for the potential cost of submitting an abbreviated application for surface sanitizers.footnote 18 For disinfectants, it is unclear how this cost would change to reflect the cost of only submitting an application form, but it is assumed to be significantly reduced as the application form is less complex and does not include providing a label text incorporating these changes. Therefore, Health Canada assumes the cost of reapplication to be $100 per disinfectant based on the itemized costs for a reapplication form previously provided by industry in response to consultations that took place for the interim order transition for COVID-19 drugs. The reapplication cost for disinfectants is considered an administrative burden to industry and is discussed further in the “One-for-one rule” section below. With 69 surface sanitizers and 1 919footnote 10 disinfectants currently on the market, the one-time cost to submit this information is expected to be $1.0 million at year five, the final year of the transition period.
Costs to Government
The total direct cost to the Government of Canada for the proposed Biocides Regulations would include the one-time cost of implementing internal systems, a one-time cost of revising standard letters, and a one-time cost of reviewing abbreviated applications received from the transition of biocides from the FDR and the PCPA. The total direct costs also include an increase in volume of post-authorization activities, an increase in requests for guidance or clarification from industry and the ongoing cost of implementing a recall and record-keeping system, which includes compliance verification activities. These costs are estimated to be $1.4 million PV or $154,848 annualized over a 15-year period.footnote 11
Transition costs
There are currently separate systems (forms, databases, internal and external applications) for the 1 919 disinfectants under the FDA and the 69 surface sanitizers under the PCPA. Health Canada maintains correspondence letter templates that are used to communicate information to disinfectant applicants throughout the submission process.
The high-level system requirements for the proposed Biocides Regulations that would apply to all disinfectants and surface sanitizers include the cost of creating and automating a common tracking system, and updating forms and standardized correspondence letter templates. In order to meet these new requirements, the current system would need to be updated accordingly. These updates will be implemented in year one, the year of the registration of the proposed Biocides Regulations, which is estimated to have a one-time cost of $503,688.
Cost of reviewing abbreviated applications for transitioning products
Under the proposed approach, Health Canada would need to review abbreviated applications for surface sanitizers and disinfectants being submitted by current registrants and authorization holders to transition to the proposed Biocides Regulations. It is assumed that Health Canada would receive and review most of the abbreviated applications within the last year of the transition period.
For surface sanitizers, it is estimated to cost Health Canada approximately $762 per surface sanitizer, with a total one-time cost of $52,606. For disinfectants, it is estimated to cost Health Canada approximately $155 per disinfectant with a total one-time cost of $297,157. The total cost of reviewing abbreviated applications for disinfectants and surface sanitizers is anticipated to be a one-time cost of $349,763 at year five, the final year of the transition period.
Ongoing government costs
Under the proposed Biocides Regulations, there would be ongoing costs to the government for reviewing documentation associated with recalls and with the increase in post-authorization applications and client service requests. Overall, these ongoing costs are anticipated to begin in year two, the year the Regulations come into force, and are estimated to result in an incremental cost of $9,606 annually.
Compliance verification
Under the proposed Biocides Regulations, the cost to government for compliance verification would grow, as a result of the increase in the number of biocides in Canada beginning in fiscal year 2023–2024. Overall, these costs are anticipated to begin in year 2 when the Regulations come into force. The compliance verification costs are estimated at $115,000 for year 2, $86,000 for year 3, $58,000 each of year 4 and year 5, $86,000 for year 6, and $58,000 per year for year 7 to year 15. The year following the end of the transition period is expected to see an increase in the number of compliance verifications because of increased activity from organizations adapting and being completely regulated under the proposed Biocides Regulations.
Qualitative costs
Increased competition for Canadian companies
The introduction of the UFD pathway is likely to encourage companies that have products that are already approved by the U.S. EPA to apply for a market authorization in Canada. This could create greater competition for market share and have an impact on the competitiveness of biocide companies that have products solely marketed in Canada. However, some Canadian companies may retain market share based on brand awareness and consumer preferences for products manufactured in Canada.
A business that chooses to apply for a market authorization through the UFD pathway would pay a lower fee and have a shortened review time than if they applied through the full review pathway. Since a reciprocal agreement would not be in place, companies that would seek approval in Canada before seeking approval in the U.S. would need to consider the order of those approvals to use the UFD pathway and access potential savings. For example, these companies would need to consider the fees and timelines associated with obtaining a product registration in the U.S. first, to ensure they would benefit from using the UFD pathway, as opposed to using an alternative Canadian application pathway.
Post-market requirements (including safety monitoring, recall reporting, quality control and record keeping)
Safety monitoring, record keeping and reporting costs would be considered a compliance cost for industry. These activities are for the purposes of ensuring a safety management system and not for demonstrating compliance to the government. Based on industry responses to the CBA survey, the potential cost to manufacturers of these proposed requirements was deemed to be cost neutral. However, for recall reporting, record keeping and quality control, the cost may vary depending on if those within the supply chain that sell a biocide have adequate systems in place. Health Canada is not aware of any distributors or importers that are non-compliant with the current guidance on maintaining a system for tracking complaints and their resolution. If a business is non-compliant, Health Canada believes it would cost them roughly $86,000 to set up a system and train staff in the first year, and then $30,000 annually thereafter. This is assuming that the type of system put in place would be similar in nature to an ISO 9001 Quality Management System and would therefore represent what Health Canada believes to be the upper bound for costs.footnote 41
Safety monitoring
Under the proposed Biocides Regulations, the MAH would have to meet the proposed safety monitoring requirements. For MAHs of surface sanitizers and disinfectants, these would be considered new requirements. However, for the CBA analysis, it is assumed that manufacturers of disinfectants and surface sanitizers already have the proposed systems in place, as it is the current recommendation in guidance.footnote 22
Recall reporting
In the event of a recall, the MAH and every person who sells a biocide, other than a retailer, would need to have a system of control in place that allows them to take effective and rapid action to recall a biocide. If they do not have a system of control in place prior to the coming into force of the proposed Regulations, this may be a potential cost to implement.
Record keeping and quality control
Those within the supply chain that sell a biocide would be required to maintain records, such as sales records and evidence of lot-specific quality control, consistent with the current practice as recommended in guidance for disinfectants and other drugs. Therefore, we assume no new costs to disinfectant manufacturers specifically. In the case that manufactures of surface sanitizers do not already maintain records consistent with these practices, there may be a potential cost to implement this. This cost has not been monetized, as there is insufficient data to assess the potential incremental cost to businesses and the number of businesses currently without the proposed systems in place.
Loss of registration value
As a result of the proposed Biocides Regulations, some sanitizer businesses may lose the value of one year of their renewal fee because of the timing of the regulatory transition period. As there are 21 surface sanitizers whose registration will expire on December 31, 2022, their renewal cycle may be cut short depending on when these products will transition to the proposed Biocides Regulations.
Benefits
The total direct benefit to industry and the Government of Canada from the proposed Biocides Regulations is estimated to be $61.6 million PV or $6.8 million annualized over a 15-year time period.
Benefits to industry
The total direct benefits to industry for the proposed Biocides Regulations include the removal of the annual summary report requirement, the introduction of the UFD pathway, and the removal of the requirement for surface sanitizers to renew their registration every five years. These benefits are estimated to be $ 60.5 million PV or $6.6 million annualized over a 15-year period.
Annual summary report cost savings
Currently for disinfectants, authorization holders must prepare and submit upon request an annual summary report to comply with safety and monitoring requirements under the FDR. Surface sanitizers registered under the PCPA are subject to annual summary reporting requirements under the Pest Control Products Incident Reporting Regulations (PCPIRR) if they have been implicated in at least 10 incidents in one year. In addition, Health Canada conducts post-market reviews of registered pest control products to confirm continued acceptability via periodic re-evaluationsfootnote 32 and special reviews.footnote 24 In both cases, the Minister can decide to continue the registration as is, require modifications (e.g. by establishing measures to mitigate risks), or cancel it.
Under the proposed Biocides Regulations, annual summary reports would no longer be required and would be replaced with new safety monitoring requirements as outlined above under the qualitative costs section. Based on industry responses during consultations and the qualitative responses received in the biocides CBA survey, the removal of annual summary reports is anticipated to have annual savings to industry. Based on cost estimates provided during consultations for the Self-Care Framework, the removal of the annual summary report requirement is estimated to save industry between $1,632 and $5,560 per product.footnote 25 The Self-Care Framework consultations were used as a proxy in this instance because the Self-Care Framework also measured the impact of removing the annual summary report with similar stakeholders. The total activities related to the preparation of the annual summary report has been estimated to require between 27 and 92 hours, with an average cost of $60 per hour. The average cost would therefore be $3,570 per product. The total savings to industry from the removal of annual summary reports would therefore be over $4.1 million.footnote 26 These cost savings would begin once registrants and authorization holders transfer their products to the proposed Regulations, which is assumed to occur in year 5, the final year of the transition period. These are considered administrative cost savings to industry and are discussed further in the “One-for-one rule” section below.
Use of foreign decisions — application pathway cost savings
Currently, most disinfectants receive a DIN under Division 1 in Part C of the FDR. Submissions for disinfectants with a new active ingredient or a new use receive an NOC and a DIN under Division 8 in Part C of the FDR. Surface sanitizers receive a pest control product registration number under the PCPA. In all cases, for new disinfectants and surface sanitizers, an application that includes evidence to demonstrate compliance with the applicable regulations must be submitted.
The proposed Biocides Regulations would introduce an additional authorization pathway for products that have been approved by a trusted foreign regulatory authority. The UFD pathway would allow applicants with an existing authorization from a foreign jurisdiction to bring their biocides to the Canadian market sooner at a reduced cost by submitting an application through this pathway.
Industry indicated through the CBA survey that obtaining a market authorization through the UFD pathway would provide a shorter review time and is estimated to save $57,850 on average per application. Based on data collected through the CBA survey, 47 applications are anticipated to come through the UFD pathway as an alternative to the existing pathway. Based on an environmental scan of biocide companies, 70% of applications will come from Canadian companies. Therefore, it is assumed these companies would benefit from the UFD pathway, and this would result in an annual cost savings of $1.9 million starting in year 2, the year the Regulations come into force.
Removing the renewal requirement for surface sanitizers
Under the current approach, registrants of surface sanitizers are required to renew their existing products by submitting an application for renewal every five years in order to continue selling their product. The cost of submitting a renewal application is considered to be significantly less than an application for a new product since it excludes data. The current fee to submit to Health Canada a renewal application for an existing product is $196.footnote 27
Based on the 94 surface sanitizers that will be on the market by 2027, that are expected to be renewed every five years, the industry is spending $18,424 every five years or $3,685 annually with the assumption that they are not all registered within the same year and are evenly distributed. Under the proposed Biocides Regulations, the renewal of surface sanitizers would no longer be a requirement and would therefore be considered a cost saving of $3,685 annually starting at year 5, the final year of the transition period.footnote 28
Benefits to Government
The total direct benefits to the Government of Canada for the proposed Biocides Regulations include the annual cost savings from the introduction of the UFD pathway and eliminating the need to review the surface sanitizer renewal requirements. The total direct benefits are estimated to be $1.1 million PV or $118,677 annualized over a 15-year period.
Review process for the use of foreign decisions pathway
Applications for disinfectants are currently submitted through Division 1 or Division 8, in Part C of the FDR, and surface sanitizer applications are submitted through the PCPA. Based on the current list of information requirements for application submissions to Health Canada, it costs Health Canada $2,248 on average to conduct a full review of an application for a disinfectant, and $7,436 on average to conduct a full review of an application for a surface sanitizer.footnote 29
Under the proposed Biocides Regulations, the Minister would have the ability to issue a market authorization by leveraging a decision issued by a foreign regulatory authority when reviewing new applications. Conducting a review of an abbreviated application through the UFD pathway is expected to save $1,387 per application.
Based on industry responses to the CBA survey, Health Canada expects to receive approximately 47 applications, on average, that would be considered eligible for the UFD pathway on an annual basis. Therefore, reviewing applications received through the UFD pathway would save Health Canada a total of $69,097 annually starting in year two, the year the Regulations come into force.footnote 30
Eliminating review of renewal applications for surface sanitizers
Currently, Health Canada processes renewal applications submitted by industry every five years for surface sanitizers once they are approved. Based on internal data, it is estimated to cost Heath Canada approximately $268 per submission, on average, to process a renewal application for a surface sanitizer. The information requirements for a renewal application are more condensed than those required for a new surface sanitizer. In addition, these applications require less time and effort to review. Assuming that renewal applications are made every five years for the estimated 94 surface sanitizers that will be on the market by 2027, this is estimated to cost Health Canada $25,192 every five years, or approximately $5,038 annually assuming the number of applications received are evenly dispersed each year.footnote 31 Under the proposed Biocides Regulations, Health Canada would no longer receive and review renewal applications for surface sanitizers following the coming into force. The Government of Canada will save $5,038 annually starting at year 5, the final year of the transition period.footnote 26
Total decrease in review process for new biocide applications
The current average cost to review an application for new products for a disinfectant can range from $607 to $2,248, depending on the application type. The cost to conduct a full review for a new surface sanitizer product is estimated to be an average of $7,436.
Under the proposed Biocides Regulations, it is assumed that there would be a minor increase in screening and review time for certain disinfectant applications, including a full review and a labelling standard review for disinfectants. However, there would be a decrease in time to conduct a full review of new surface sanitizers. The overall net review time for all biocides is expected to decrease, and this is expected to be a cost savings of $9,876 annually. These annual savings are expected to begin in year two, the year the Regulations come into force.footnote 26 footnote 23
Overall, when accounting for all monetized costs and benefits, a $59 million PV or $6.5 million annualized net benefit is anticipated over the next 15 years applying a 7% discount rate.
Qualitative benefits
Ability to include multiple brand names in a market authorization
The proposed Biocides Regulations would introduce the ability for one market authorization to include multiple brand names or multiple versions of the biocide. This would have cost savings for industry and Government, as it would allow for a more efficient application submission and review process. This is anticipated to reduce burden to both industry and the Government of Canada.
Increased supply of biocides to provide more timely access to Canadians
The COVID-19 pandemic has created an unprecedented demand on Canada’s health care system. The need for health products, including disinfectants and surface sanitizers, has exponentially increased. The proposed Biocides Regulations would introduce a UFD application pathway that would remove barriers to both Health Canada and industry, providing Canadians with more timely access to biocides. Many Canadian biocide suppliers have parent companies in other countries, predominantly in the United States. The industry includes not only established large companies, but also small and medium-sized companies, bringing innovative products to global markets. Through the proposed UFD pathway, international collaboration may promote the timely approval of a variety of safe and effective biocides. For the past several years, Health Canada has leveraged data from other regulatory agencies, when applicable, in order to better inform the review of biocides for market authorization in Canada. By proposing to expand this practice, it will be of benefit to Canadians by helping to ensure a safe, effective, and high quality supply of biocides is maintained.
Increased variety of biocides for Canadians
As a result of the proposed Biocides Regulations, Canadians would have timely access to a larger variety of biocides. With the potential increase in additional products expected through the UFD pathway, it is anticipated to be an impact on product pricing and competition in the market that would benefit Canadians.
Cost-benefit statement
- Number of years: 15 years (2023 to 2037)
- Base year for costing: 2021
- Present value base year: 2023
- Discount rate: 7%
| Impacted stakeholder | Description of cost | Base year (2023) | Other relevant year (2024) |
Other relevant year (2027) |
Final year (2037) | Total (present value) | Annualized value |
|---|---|---|---|---|---|---|---|
| Industry | Label change | $0 | $0 | $566,490 | $0 | $403,900 | $44,346 |
| Reapplication | $0 | $0 | $1,019,900 | $0 | $727,175 | $79,840 | |
| Government | Transition costs | $503,688 | $0 | $0 | $0 | $470,736 | $51,684 |
| Cost of reviewing abbreviated applications for transitioning products | $0 | $0 | $349,763 | $0 | $249,376 | $27,380 | |
| Ongoing costs | $0 | $9,606 | $12,101 | $26,125 | $124,873 | $13,710 | |
| Compliance verification | $0 | $115,000 | $58,000 | $58,000 | $565,353 | $62,073 | |
| All stakeholders | Total costs | $503,688 | $124,606 | $2,006,254 | $84,125 | $2,541,413 | $279,033 |
| Impacted stakeholder | Description of benefit | Base year (2023) |
Other relevant year (2024) |
Other relevant year (2027) |
Final year (2037) |
Total (present value) | Annualized value |
|---|---|---|---|---|---|---|---|
| Industry | Annual summary report cost savings | $0 | $0 | $4,134,060 | $8,925,125 | $33,981,172 | $3,730,950 |
| UFD pathway application cost savings | $0 | $1,903,259 | $2,397,559 | $5,176,149 | $26,473,259 | $2,906,622 | |
| Removing requirement for renewal (surface sanitizers) | $0 | $0 | $3,685 | $7,956 | $39,704 | $4,359 | |
| Government | Review process for UFD | $0 | $69,097 | $87,043 | $187,919 | $898,230 | $98,621 |
| Eliminating review of renewal applications for surface sanitizers | $0 | $0 | $5,038 | $10,877 | $54,282 | $5,960 | |
| Total decrease in review process for new biocide applications | $0 | $9,876 | $12,441 | $26,860 | $128,385 | $14,096 | |
| All stakeholders | Total benefits | $0 | $1,982,233 | $6,639,826 | $14,334,886 | $61,575,032 | $6,760,608 |
| Impacts | Base year (2023) |
Other relevant year (2024) |
Other relevant year (2027) |
Final year (2037) |
Total (present value) | Annualized value |
|---|---|---|---|---|---|---|
| Total costs | $503,688 | $124,606 | $2,006,254 | $84,125 | $2,541,413 | $273,033 |
| Total benefits | $0 | $1,982,233 | $6,639,826 | $14,334,886 | $61,575,032 | $6,760,608 |
| NET IMPACT | −$503,688 | $1,857,627 | $4,633,572 | $14,250,761 | $59,033,619 | $6,481,574 |
Quantified (non-$) and qualitative impacts
Positive impacts
- Increased predictability in application requirements for similar products would help streamline the filing of applications for multiple products;
- Ability to allow for one market authorization to include multiple brand names or multiple versions of the biocide would lead to cost savings for both industry stakeholders and the Government of Canada;
- Facilitating and maintaining timely access to a larger variety and supply of biocides would address the need for consumer and commercial product choice; and
- Would allow Health Canada to request information about a biocide from an MAH when a concern is brought to its attention.
Negative impacts
- The proposal does not include disinfectants and sanitizers that are used for different methods of application (e.g. air and water);
- The UFD pathway may introduce increased competition for existing Canadian companies and may impact their profits; and
- The UFD pathway may present a disadvantage to Canadian firms that do not wish to enter the U.S. market first, as they would not benefit from the UFD pathway.
Small business lens
As of 2021, there are approximately 230 small businesses that have disinfectants and surface sanitizers authorized for sale in Canada. Based on a sampling of companies in Health Canada’s database, an average small business has 1.9 products that would be regulated under the proposed Biocides Regulations. Therefore, small businesses account for approximately 437 products. Assuming that the proportion of surface sanitizers to disinfectants is the same for small businesses as the proportion of these products on the total Canadian market, small businesses would have 422 disinfectants and 15 surface sanitizers authorized for sale in Canada.
The submission of an abbreviated application to transition disinfectants would cost small businesses $42,200, assuming 422 products and $100 cost per product.
The cost to submit an abbreviated application to transition surface sanitizers is estimated to be $12,000, assuming that small businesses produce 15 surface sanitizers, for a total cost of $180,000.
The total cost for small businesses to physically change their surface sanitizers’ labels during the four-year transition period is estimated to be $123,150. This is assuming 15 products at a cost of $7,608, where half may need to make label amendments at a cost of $1,204 each (to continue uses that are regulated under the PCPA) in year five, the final year of the transition period. A shorter or longer transition period would change the cost per label. Health Canada considered the concerns of small businesses and concluded that a transition period of four years would help alleviate some of the burden for both large enterprises and small and medium-size enterprises.
All businesses would benefit from reduced requirements around post-market reporting, including small businesses. Assuming small businesses account for approximately 22% of all biocides (437 products), these businesses are expected to save the following amounts over the next 15 years:footnote 33
- $740,782 annually in annual report savings;
- $949 annually with the removal of the requirement to submit renewal applications for surface sanitizers every five years; and
- $774,975 annually with the introduction of the UFD pathway.
Taken over a 15-year period, with some of the benefits starting in year 5, discounted at 7%, this would save small businesses approximately $10.6 million in PV or $1.5 million annually.footnote 34
Overall, the net benefit to small businesses would be $10.4 million in PV over 15 years using a 7% discount rate. Divided by 230 small businesses, this creates a net benefit of $45,246 or $6,442 annually per small business.
The introduction of the UFD pathway may encourage small businesses to expand by accessing foreign products to sell in Canada. Small businesses that tend to introduce products onto both the Canadian and U.S. markets may take advantage of the cost savings under the UFD. Small businesses that have sought Canadian market access first may change their business practice for subsequent biocide applications and seek U.S. approval first as a result of the UFD pathway. In addition, small businesses that previously did not seek or intend to seek U.S. approval may choose to change their approach.
Small businesses that do not intend to market in the United States may find that the Canadian market would be more competitive as a result of the proposed Regulations, reducing margins and thereby profits. These reduced margins would also mean that any additional costs from the proposed Biocides Regulations may be passed on to consumers by small business, since the ability for these businesses to absorb additional costs in the short term is less likely.
The 2020 fee update for drugs, including disinfectants, introduced small business fee reduction measures that would be maintained for biocides in the accompanying fee proposal. These small business fee reduction measures would help to protect the interests of small businesses.
Small business lens summary
- Number of small businesses impacted: 230
- Number of years: 15 (2023–2037)
- Base year for costing: 2021
- Present value base year: 2023
- Discount rate: 7%
| Activity | Annualized value table c4 note a | Present value |
|---|---|---|
| Updating labels | $12,501 | $87,804 |
| Reapplication — Surface sanitizers | $18,272 | $128,338 |
| Removing requirement for renewal (sanitizers) | −$949 | −$6,664 |
| UFD pathway | −$774,975 | −$5,443,100 |
| Total compliance benefit | $745,151 | $5,233,622 |
Table c4 note(s)
|
||
| Activity | Annualized value | Present value |
|---|---|---|
| Reapplication — Disinfectants | $4,284 | $30,088 |
| Elimination of annual summary reports | −$740,782 | −$5,202,940 |
| Total administrative benefit | $736,498 | $5,172,852 |
| Totals | Annualized value | Present value |
|---|---|---|
| Total cost (all impacted small businesses) | $35,058 | $246,230 |
| Cost per impacted small business table c6 note a | $152 | $1,071 |
Table c6 note(s)
|
||
One-for-one rule
For the purposes of the one-for-one rule, the proposed Regulations would be a title “in,” that is to say the proposal creates a new regulatory title under the FDA. For the purposes of counting administrative burden, the proposed Regulations would be an “out.”
Reapplication costs for disinfectants and surface sanitizers
Under the Policy on Limiting Regulatory Burden on Business, pre-market approval processes are considered a compliance cost. The transfer of surface sanitizers to the proposed Biocides Regulations under the FDA and the requirement to submit abbreviated applications for surface sanitizers for approval would not be considered an administrative burden under the one-for-one rule. These abbreviated applications for surface sanitizers would be used to expedite approval from one regime (PCPR) to another (FDA) and would fall under the pre-market approval process.
Administrative costs — abbreviated application to transition disinfectants
Under the proposed Biocides Regulations, authorization holders of disinfectants must transition their biocide by filing an abbreviated transition application to Health Canada if they plan to continue the sale of these products as biocides. The abbreviated application filed to Health Canada by existing disinfectant authorization holders currently operating under the FDR would be considered a one-time administrative cost. This one-time cost is estimated to be $100 in 2021 dollars ($83 in 2012 dollars) per application, or $191,900 in total.
Administrative cost savings: Removal of annual summary report
The reduced cost of post-market reporting as a result of removing the annual summary report requirement for biocide MAHs would result in a cost savings. The removal of the requirement to submit an annual summary report has been estimated by industry to save between $1,632 and $5,560 per product in 2021 dollars ($1,386 and $4,721 in 2012 dollars respectively).footnote 35 The activities related to the preparation of the annual summary report has been estimated by industry to require between 27 and 92 hours at an average cost of $60 per hour.
| Description of costs | Values to report in regulatory impact analysis statement table c7 note * |
|---|---|
| Annualized administrative costs | −$1,528,439 |
| Annualized administrative costs per business | −$482.31 |
Table c7 note(s)
|
|
Regulatory cooperation and alignment
The proposed Regulations would allow for the authorization and regulation of biocides under a single framework and separately from other health products under the FDA and pest control products under the PCPA. This would be consistent with the approach taken by the United States, the U.K. and the EU. The regulatory requirements for biocides regulated by the U.S. EPA are able to meet those in the proposed Biocides Regulations (e.g. requirements related to the application structure, recall, record keeping, and packaging specifications).
Following initial consultation with industry stakeholders on the proposed Biocides Regulations, Health Canada further aligned proposed application requirements with those of the United States, the U.K. and the EU, by allowing one application to be submitted for multiple products with minor variations in formulation and brand names.
The UFD pathway in the proposed Biocides Regulations is based on the confidence that Health Canada has in other foreign regulators as a result of collaboration, harmonization and other regulatory confidence-building activities. The intent of this pathway is to promote trade and further alignment with other jurisdictions to promote access to a larger variety of safe and effective biocides for Canadians. Recently, the U.S. EPA has been making joint efforts with Health Canada to streamline requirements for pest control product submissions. In addition, the concept of using foreign decisions and similar models in product evaluation and assessment has been adopted for therapeutics and pharmaceuticals by the regulatory authorities in Australia, Switzerland and Singapore. It is to be noted that certain differences exist between the requirements in Canada and other international jurisdictions. These are necessary to align with the structure and requirements in other applicable Acts and regulations in Canada, and to align with Canada’s bilingual labelling requirements.
Strategic environmental assessment
As there are new labelling requirements proposed through the Biocides Regulations, it is anticipated that labels of all surface sanitizers would be required to change to come into compliance with the proposed Regulations. The proposed Biocides Regulations would come into force one year after they are registered. At that point, all new products entering the market would have to meet the proposed requirements. Those who hold a registration for a surface sanitizer under the PCPA or an authorization for a disinfectant under the FDR would have a transition period of four years from the coming-into-force date to obtain an authorization under the proposed Biocides Regulations. During this time frame, some companies would engage in some form of relabelling at a time most convenient for their business. This would minimize costs on industry and avoid the risk of supply chain bottlenecks with third party designers, labellers, and packagers. This transition strategy would therefore minimize the need for biocides to be recalled, repackaged, discarded, or relabelled, to comply with the proposal. The transition plan would also help minimize waste by enabling MAHs to sell off existing stock, while still requiring that all products come into compliance with the proposed requirements as quickly and efficiently as possible.
Gender-based analysis plus
Health Canada expects that the proposed Biocides Regulations would have a positive impact on Canadians.
Incidents
Certain vulnerable populations may be more prone to incidents from the misuse of, overuse of, or accidental poisoning from biocides. According to data from poison control centres in the United States, exposures among children aged five years or younger consistently represented a large percentage of total reports throughout the three-month period for each year that was studied (January to March 2018, 2019, and 2020).footnote 36,footnote 37 Similar findings were reported from Canadian poison control centres.footnote 38 To help mitigate this, the proposal would require the statement ‘‘Keep out of reach of children’’ on the principal display panel of all biocides to help prevent any unintentional exposure by this subpopulation. In addition, Health Canada is proposing to include application requirements that relate to the packaging of the biocide to prevent the use of food like- and beverage-like packaging. Appropriate safety labelling statements would continue to be a regulatory requirement to mitigate any risks associated with biocides.
Workplace-related exposure
There may be greater occupational exposure to biocides due to the nature of certain professions. For example, people in health care, food preparation, livestock, childcare and janitorial service industries are likely to have greater exposure to these types of products, as the use of biocides in these settings is necessary to prevent microbial growth and outbreaks.footnote 39 There may be increased adverse effects observed for these population groups, such as allergic responses,footnote 40 or chronic obstructive pulmonary disease as a result of this increased occupational exposure.footnote 21
The proposal requires that the MAH of each biocide would be required to monitor the safety of their product over time in a manner that reliably allows them to detect significant safety issues or changes to the risks and benefits associated with the product that emerge as a result of real-world use. The Minister would have the authority to compel label changes, recall a product, request information from an authorization holder, suspend or revoke a product authorization and direct persons to stop the sale to mitigate risks, if necessary.
Implementation, compliance and enforcement, and service standards
Implementation
Health Canada would be publishing communication materials and hosting information sessions before the coming into force to facilitate the understanding of the proposed Biocides Regulations. Communications would clearly outline the transition process for surface sanitizers regulated under the PCPA and disinfectants regulated under the FDR. In addition, formal guidance related to each aspect of the proposed Biocides Regulations would be made available to stakeholders should the Regulations be published in the Canada Gazette, Part II.
Health Canada would issue stakeholder communications to encourage registration and authorization holders to transition their products before the end of the transition period.
Health Canada would regularly review the List of Foreign Authorities associated with the UFD pathway to provide opportunities to add or remove trusted regulatory authorities or application pathways. Health Canada would consult with Canadians as required through a notice of proposal published on the Department’s website prior to implementing changes to the List of Foreign Authorities.
It is anticipated that the cost associated with the review of an application submitted through the proposed UFD pathway would be less than that associated with a full review. Health Canada is consulting on a fee proposal along with the pre-publication of the proposed Biocides Regulations.
Compliance and enforcement
As part of its regulatory responsibilities, Health Canada monitors compliance, undertakes enforcement actions, and works towards preventing non-compliance through compliance promotion activities. Health Canada would extend its current Compliance and enforcement policy for health products (POL-0001) to include biocides. Any impacted quality documents would be updated prior to the coming into force of the proposed Biocides Regulations.
Currently, surface sanitizers and disinfectants are not subject to requirements of the FDR related to drug establishment licensing and good manufacturing practices. Health Canada conducts compliance and enforcement activities for these products using a combination of compliance monitoring, compliance promotion, and a risk-based approach to inspection and compliance verification that is typically triggered by international monitoring, identified quality issues, or complaints. This risk-based compliance and enforcement approach would continue to apply for biocides under the proposed Regulations.
Service standards
Health Canada would establish service standards for biocides according to the level of effort required to review a product application and issue a decision. Health Canada is consulting on proposed fee lines and service standards along with the prepublication of the proposed Biocides Regulations.
Performance results would be published each fiscal year in the Report on Fees, which is tabled in Parliament in the following fiscal year.
Contact
Bruno Rodrigue
Executive Director
Office of Legislative and Regulatory Modernization
Policy, Planning and International Affairs Directorate
Health Products and Food Branch
Health Canada
Holland Cross, Suite P2108
11 Holland Avenue
Ottawa, Ontario
K1A 0K9
Address locator: 3000A
Email: lrm.consultations-mlr@hc-sc.gc.ca
PROPOSED REGULATORY TEXT
Notice is given that the Governor in Council, pursuant to section 30footnote a of the Food and Drugs Actfootnote b and subsection 67(1)footnote c of the Pest Control Products Actfootnote d, proposes to make the annexed Biocides Regulations.
Interested persons may make representations concerning the proposed Regulations within 70 days after the date of publication of this notice. All such representations must be submitted online on the Canada Gazette, Part I, or, if submitted by email, post or other format, must cite the Canada Gazette, Part I, and the date of publication of this notice, and be addressed to Bruno Rodrigue, Executive Director, Office of Legislative and Regulatory Modernization, Health Products and Food Branch, Department of Health, Address Locator: 3000A, 11 Holland Avenue, Suite P2108, Ottawa, Ontario K1A 0K9 (email: lrm.consultations-mlr@hc-sc.gc.ca).
Ottawa, May 2, 2022
Wendy Nixon
Assistant Clerk of the Privy Council
Biocides Regulations
Interpretation
Definitions
1 (1) The following definitions apply in these Regulations.
- Act
- means the Food and Drugs Act. (Loi)
- active ingredient
- means a component of a biocide that is directly responsible for any of the biocide’s intended effects. (ingrédient actif)
- biocide
- means a drug that is sold or represented for use in destroying or inactivating micro-organisms, or in reducing or controlling their number, on a non-living and non-liquid surface. However, it does not include such a drug that is sold or represented for use
- (a) on the surface of an invasive device as defined in section 1 of the Medical Devices Regulations; or
- (b) exclusively on the surface of food. (biocide)
- CAS registry number
- means the identification number that is assigned to a chemical substance by the Chemical Abstracts Service division of the American Chemical Society. (numéro d’enregistrement CAS)
- conditions of use,
- in respect of a biocide, means
- (a) its intended uses or purposes;
- (b) its intended users;
- (c) the settings in which it is intended to be used;
- (d) its risk information;
- (e) the directions for use; and
- (f) the directions for storage and disposal. (conditions d’utilisation)
- contact information
- includes civic address, except in paragraphs 31(1)(e), 48(2)(d) and 49(1)(a). (coordonnées)
- expiry date,
- in respect of a biocide, means the year and month in which its shelf life expires. (date limite d’utilisation)
- foreign regulatory authority
- means a government agency or other entity outside Canada that controls the manufacture, sale or use of biocides within its jurisdiction and that may take enforcement action to ensure that biocides marketed within its jurisdiction comply with the applicable legal requirements. (autorité réglementaire étrangère)
- formulant
- means any component of a biocide other than an active ingredient or contaminant. (formulant)
- import
- means import for the purpose of sale. (importer)
- List of Foreign Regulatory Authorities
- means the document entitled List of Foreign Regulatory Authorities for Biocides, published by the Government of Canada, as amended from time to time. (Liste des autorités réglementaires étrangères)
- lot number
- means any combination of letters, figures or both by which a biocide can be traced in manufacture and identified in distribution. (numéro de lot)
- master formula,
- in respect of a biocide, means a document that sets out
- (a) the ingredients that are used in the manufacture of the biocide and the quantities of those ingredients that are required to manufacture a given quantity of the biocide;
- (b) the specifications for the biocide;
- (c) a detailed description of the procedures required to manufacture, package, label and store the biocide, including safety precautions and in-process controls;
- (d) a detailed description of the methods used to test and examine the packaging material; and
- (e) a statement of tolerances for the properties and qualities of the packaging material. (formule type)
- shelf life
- means the period, beginning on the day on which a biocide is packaged for sale to consumers, during which the biocide will, when stored appropriately, retain without any appreciable deterioration its stability and any other qualities claimed for it by the holder of the market authorization for it. (durée de conservation)
- specifications,
- in respect of a biocide, means
- (a) a statement of all of its properties and qualities, and of all of the properties and qualities of its active ingredients and formulants, including the identity, potency and purity of the biocide and those ingredients and formulants;
- (b) the size and type of its package and a statement of all of the properties and qualities of the packaging material;
- (c) a detailed description of the methods used to test and examine the biocide and its active ingredients and formulants; and
- (d) a statement of tolerances for the properties and qualities of the biocide and for the properties and qualities of its active ingredients and formulants. (spécifications)
Definition of serious adverse drug reaction
(2) For the purposes of section 21.8 of the Act as it relates to biocides, serious adverse drug reaction means a response to a biocide that, in respect of human health,
- (a) results in in-patient hospitalization, prolongation of existing hospitalization, congenital malformation or chronic or significant disability or incapacity; or
- (b) is life-threatening or results in death.
Non-application
Food and Drug Regulations
2 Except as otherwise provided in these Regulations, the Food and Drug Regulations do not apply to a biocide.
Import and Export
Prohibition — import of non-compliant biocides
3 (1) Subject to subsection (2), it is prohibited to import a biocide if its sale in Canada would contravene the Act or these Regulations.
Exception
(2) The prohibition does not apply if
- (a) the sale of the biocide in Canada would not contravene the Act or these Regulations if the biocide was relabelled or modified; and
- (b) the importer provides an inspector with advance notice of the import.
Prohibition — importer information not published
4 It is prohibited to import a biocide unless the importer’s name and civic address have been published by the Minister in connection with the biocide under subsection 11(2).
Export certificates
5 (1) For the purposes of paragraph 37(1)(c) of the Act, an export certificate in respect of a biocide must be in the form set out in Appendix III to the Food and Drug Regulations.
Obligation to retain copy
(2) The exporter must retain a copy of the export certificate for at least five years after the day on which the biocide is exported.
Exemptions — Section 3 of the Act
Advertising as preventative only
6 A biocide is exempt from the application of subsection 3(1) of the Act if the biocide is not advertised to the general public as a treatment or cure for any of the diseases, disorders or abnormal physical states referred to in Schedule A.1 to the Act.
Sale as preventative only
7 A biocide is exempt from the application of subsection 3(2) of the Act with respect to its sale by a person if
- (a) the biocide is not represented by label as a treatment or cure for any of the diseases, disorders or abnormal physical states referred to in Schedule A.1 to the Act; and
- (b) the person does not advertise the biocide to the general public as a treatment or cure for any of those diseases, disorders or abnormal physical states.
Market Authorizations
Prohibition
Import, sale or advertising
8 (1) Subject to subsection (2), it is prohibited to import, sell or advertise a biocide unless the biocide is the subject of a market authorization issued under section 10.
Exception — testing
(2) Subsection (1) does not prohibit the sale of a biocide for the purpose of testing to support an application for a market authorization for that biocide under section 9.
Issuance
Application requirements
9 (1) An application for a market authorization for a biocide must be submitted to the Minister and contain
- (a) the applicant’s name and contact information;
- (b) the brand names under which the biocide is proposed to be sold or advertised;
- (c) the physical form of the biocide and the methods of application;
- (d) a list of the biocide’s active ingredients and their CAS registry numbers, if they have one;
- (e) a list of the formulants that are contained in the biocide, including formulants that are contained in a particular version of the biocide only, and their CAS registry numbers, if they have one;
- (f) the quantity of each active ingredient and formulant that is contained in a given quantity of the biocide;
- (g) the biocide’s conditions of use;
- (h) information regarding the benefits and risks associated with the biocide and any uncertainties relating to those benefits and risks, including summaries and reports of any tests and studies conducted by or on behalf of the applicant to identify and assess those benefits, risks and uncertainties;
- (i) a summary of the information provided under paragraph (h);
- (j) the names and contact information of
- (i) the persons that will manufacture the biocide, excluding individuals who are employees or agents of such persons,
- (ii) any persons that will import the biocide, and
- (iii) if known, the persons that will package or label the biocide, excluding individuals who are employees or agents of such persons;
- (k) the civic addresses of the locations where the biocide will be manufactured and, if known, packaged and labelled;
- (l) the specifications for the biocide;
- (m) the shelf life of the biocide;
- (n) the text of every label to be used in connection with the biocide, including any supplementary information on the use of the biocide that is to be provided to users on request;
- (o) the procedure to be followed for decontamination, if any;
- (p) if the biocide is a pest control product, as defined in subsection 2(1) of the Pest Control Products Act, that is registered under that Act, the registration number; and
- (q) if the biocide is a drug for which a drug identification number has been assigned under Part C of the Food and Drug Regulations, the drug identification number.
Application based on comparison
(2) The information referred to in paragraphs (1)(h), (i) and (l), other than information relating to the size and type of the package and the properties and qualities of the packaging material, may be omitted from the application if
- (a) the application is based on a comparison between the biocide and another biocide that is the subject of a market authorization;
- (b) the application contains information that demonstrates that
- (i) the biocide contains, in comparison to the other biocide, the same active ingredients in the same quantities in a given quantity of biocide,
- (ii) the formulants contained in the biocide are among the formulants that are permitted to be contained in the other biocide under its market authorization,
- (iii) the quantity of each formulant contained in a given quantity of the biocide is the same as the quantity of that formulant that is permitted to be contained in the same given quantity of the other biocide,
- (iv) subject to subparagraph (v), the conditions of use of the biocide fall within the conditions of use of the other biocide,
- (v) the biocide has the same risk information and directions for storage and disposal as the other biocide, and
- (vi) the biocide has the same decontamination procedure as the other biocide, if the other biocide has such a procedure;
- (c) the application contains an attestation by an individual who has authority to bind the applicant confirming that the biocide will be manufactured in accordance with the master formula for the other biocide;
- (d) the market authorization for the other biocide was not issued on the basis of a comparison to a third biocide and is not suspended; and
- (e) in the case where the other biocide was exempted from the application of these Regulations under subsection 63(1), the drug identification number assigned to that biocide under the Food and Drug Regulations was not assigned on the basis of a comparison to a third biocide.
Additional information and material
(3) The Minister may request the applicant to provide any additional information or any material, including samples, that is necessary to enable the Minister to determine whether the market authorization must be issued.
Obligation to issue
10 (1) The Minister must issue a market authorization for a biocide to an applicant if
- (a) the application meets the applicable requirements set out in section 9 or 25, as the case may be;
- (b) the applicant has provided the Minister with any additional information and material that is requested under subsection 9(3) or 25(5);
- (c) subject to subsection (2), the Minister has sufficient evidence to support the conclusion that the benefits associated with the biocide outweigh the risks associated with it, taking into account any uncertainties relating to the benefits and risks; and
- (d) the Minister has reasonable grounds to believe that the biocide will be packaged and labelled in accordance with the Act and these Regulations.
Exception
(2) Paragraph (1)(c) does not apply if the conditions set out in paragraphs 9(2)(a) to (e) are met.
Content of market authorization
11 (1) A market authorization for a biocide must set out
- (a) the identification number assigned by the Minister to the biocide;
- (b) the name and civic address of the holder of the authorization and the names and civic addresses of any importers of the biocide;
- (c) the biocide’s brand names;
- (d) the physical form of the biocide and the methods of application;
- (e) a list of the biocide’s active ingredients and formulants, including their CAS registry numbers, if they have one;
- (f) the quantity of each active ingredient and formulant that is contained in a given quantity of the biocide;
- (g) the biocide’s conditions of use;
- (h) the shelf life of the biocide;
- (i) the terms and conditions of the authorization, if any;
- (j) the date on which the authorization is issued;
- (k) the dates, if any, on which the authorization is amended;
- (l) in the case of an authorization issued on the basis of an application that meets the requirements set out in subsection 9(2), the identification number of the biocide that forms the basis for the comparison;
- (m) in the case of an authorization issued on the basis of an application submitted under section 25,
- (i) a statement indicating that the sale of the biocide has been authorized on the basis of a foreign decision,
- (ii) the name of the relevant foreign regulatory authority, and
- (iii) a description of the foreign biocide; and
- (n) in the case where the biocide has or had a drug identification number assigned to it under the Food and Drug Regulations on the basis of a comparison to another biocide, the drug identification number assigned to that other biocide under those Regulations.
Public notice
(2) The Minister must, after issuing a market authorization, publish the information referred to in subsection (1), except for the quantity of the formulants that are contained in a given quantity of the biocide and the shelf life of the biocide.
Information not confidential business information
(3) Any information referred to in subsection (1) that is confidential business information ceases to be confidential business information when the Minister issues the market authorization.
Terms and Conditions
Terms and conditions
12 (1) The Minister may, at any time, impose terms and conditions on a market authorization, or amend them, after considering
- (a) whether there are significant uncertainties relating to the benefits or risks associated with the biocide to which the authorization relates;
- (b) whether the requirements under the Act are sufficient to
- (i) optimize the benefits and minimize the risks associated with the biocide,
- (ii) manage the uncertainties relating to the benefits and risks, and
- (iii) collect information to be able to continuously assess the benefits and risks, identify any changes to them and manage the uncertainties; and
- (c) whether the terms and conditions that the Minister intends to impose may contribute to meeting the objectives set out in subparagraphs (b)(i) to (iii).
Obligation to amend
(2) If, after issuing a market authorization, the Minister imposes terms and conditions on the authorization or amends them, the Minister must amend the information that is published under subsection 11(2) accordingly.
Changes
Deemed creation of new biocide
13 The holder of the market authorization for a biocide that makes any of the following changes in respect of the biocide is considered to have created a new biocide that is not authorized to be imported, sold or advertised under the market authorization:
- (a) the substitution, addition or removal of an active ingredient;
- (b) a change to the quantity of an active ingredient that is contained in a given quantity of the biocide; or
- (c) a change to the physical form of the biocide.
Definition — major change
14 (1) For the purposes of this section and section 15, major change means a change, other than a change referred to in section 13, that relates to the information or material provided to the Minister in connection with a biocide and that is reasonably expected to have a major impact on the quality of the biocide, the benefits or risks associated with the biocide or the uncertainties related to the benefits and risks, including
- (a) a change to the methods of application;
- (b) the addition of a formulant, except when it is replacing a similar formulant in a similar quantity in a given quantity of the biocide;
- (c) the removal of a formulant, except when it is being replaced by a similar formulant in a similar quantity in a given quantity of the biocide;
- (d) a change to the quantity of a formulant that is contained in a given quantity of the biocide;
- (e) a change to the conditions of use, except
- (i) the removal of an intended use or purpose,
- (ii) the addition of a class of individuals with relevant training or expertise as intended users,
- (iii) the removal of a setting in which the biocide is intended to be used,
- (iv) the addition of risk information,
- (v) the addition of a direction for storage, or
- (vi) a change to the directions for disposal;
- (f) a change to the specifications for the biocide other than a change that narrows the acceptable range of variation in the potency of an active ingredient;
- (g) an extension of the shelf life;
- (h) a reduction in the shelf life if the new shelf life is less than one year; and
- (i) a change to the package that is likely to affect the shelf life of the biocide.
Prohibition
(2) It is prohibited for the holder of the market authorization for a biocide to sell any version of the biocide that is affected by a major change unless the Minister has provided the holder with a notice of acceptance in respect of the change under subsection 15(1).
Content of application
(3) An application for a notice of acceptance must be submitted to the Minister and contain
- (a) a description of the major change;
- (b) subject to subsection (4), the information referred to in subsection 9(1) that is relevant to the change; and
- (c) if the conditions set out in paragraphs (4)(a) to (c) are met,
- (i) evidence of the authorization provided by the relevant foreign regulatory authority in respect of the change, and
- (ii) an attestation by an individual who has authority to bind the holder of the market authorization confirming that the holder possesses or has immediate access to the information that was submitted to the foreign regulatory authority to obtain the authorization referred to in subparagraph (i).
Conditions — foreign regulatory authority
(4) The information referred to in paragraph (3)(b) may be omitted from the application if
- (a) the market authorization for the biocide was issued on the basis of an application submitted under section 25;
- (b) the foreign regulatory authority named in the market authorization has authorized the major change in respect of the foreign biocide described in the market authorization, other than on the basis of a type of application that is set out in the List of Foreign Regulatory Authorities in connection with that regulatory authority; and
- (c) the name of the foreign regulatory authority appears in the List of Foreign Regulatory Authorities when the application is submitted.
Additional information and material
(5) The Minister may request the holder to provide any additional information or any material, including samples, that is necessary to enable the Minister to determine whether the notice of acceptance must be provided.
Notice of acceptance
15 (1) The Minister must provide the holder of the market authorization for a biocide with a notice of acceptance in respect of a major change if
- (a) the Minister has received an application in respect of the change that contains the relevant information referred to in subsection 14(3);
- (b) the holder has provided the Minister with any additional information and material requested under subsection 14(5);
- (c) the Minister has sufficient evidence to support the conclusion that the benefits associated with the biocide outweigh the risks associated with it, taking into account any uncertainties relating to the benefits and risks; and
- (d) in the case where the change affects the packaging or labelling of the biocide, the Minister has reasonable grounds to believe that the biocide will be packaged and labelled in accordance with the Act and these Regulations.
Obligation to amend
(2) If the Minister provides the holder with a notice of acceptance in respect of a major change that relates to any information that must be set out in the market authorization, the Minister must
- (a) amend the authorization to reflect the change; and
- (b) if applicable, amend the information that is published under subsection 11(2) accordingly.
Information not confidential business information
(3) Any information that is required to be set out in the market authorization and that is confidential business information ceases to be confidential business information when the Minister provides the notice of acceptance to the holder.
Definition — minor change
16 (1) For the purposes of this section, minor change means a change that relates to the information or material provided to the Minister in connection with a biocide and that is reasonably expected to have a minor impact on the quality of the biocide, the benefits or risks associated with the biocide or the uncertainties related to the benefits and risks, including
- (a) a change to the name or contact information of the holder of the market authorization for the biocide;
- (b) a change to the biocide’s brand names;
- (c) the replacement of a formulant with a similar formulant, in a similar quantity, in a given quantity of the biocide;
- (d) a change referred to in any of subparagraphs 14(1)(e)(i) to (vi);
- (e) a change to the specifications for the biocide that narrows the acceptable range of variation in the potency of an active ingredient;
- (f) the addition, replacement or removal of an importer of the biocide or of a person that manufactures, packages or labels the biocide or a change to the name or contact information of such an importer or person;
- (g) a change in the location where the biocide is manufactured, packaged or labelled;
- (h) a reduction in the shelf life of the biocide if the new shelf life is at least one year;
- (i) a change to the package that is unlikely to affect the shelf life of the biocide; and
- (j) a change to the procedure to be followed for decontamination.
Written description
(2) Subject to subsection (3), the holder of the market authorization for a biocide must provide the Minister with a written description of any minor change in respect of the biocide at least 30 days before making the change or, if the change is not within the holder’s control, as soon as feasible after the holder becomes aware of the change.
Names and contact information
(3) The holder must inform the Minister in writing as soon as feasible if there is a change to their name or contact information or a change referred to in paragraph (1)(f).
Obligation to amend
(4) If the minor change relates to any information that must be set out in the market authorization, the Minister must
- (a) amend the authorization to reflect the change; and
- (b) if applicable, amend the information that is published under subsection 11(2) accordingly.
Information not confidential business information
(5) Any information that is required to be set out in the market authorization and that is confidential business information ceases to be confidential business information when the holder provides the description of the minor change to the Minister under subsection (2).
Direction to Stop Sale
Prohibition
17 It is prohibited for a person to sell a lot or batch of a biocide if they have been directed to stop the sale of the lot or batch under subsection 18(1) and the direction has not been lifted.
Direction to stop sale
18 (1) The Minister may direct a person to stop the sale of a lot or batch of a biocide if the Minister has reasonable grounds to believe that
- (a) the lot or batch is not manufactured, packaged or labelled in accordance with the master formula for the biocide or has not been stored in accordance with the master formula; or
- (b) the lot or batch is manufactured, packaged or labelled in contravention of the Act or these Regulations.
Obligation to lift direction
(2) The Minister must lift a direction if the Minister is provided with information that demonstrates, or the Minister determines, that
- (a) in the case of a direction made for the reason set out in paragraph (1)(a), the lot or batch is manufactured, packaged or labelled, or has been stored, as the case may be, in accordance with the master formula for the biocide; or
- (b) in the case of a direction made for the reason set out in paragraph (1)(b), the lot or batch is manufactured, packaged or labelled, as the case may be, in accordance with the Act or these Regulations, as the case may be.
Suspension
Prohibition — sale or advertising
19 (1) Subject to subsection (2), it is prohibited for a person to sell or advertise a biocide if the market authorization for the biocide is suspended under section 20 and the person has been notified of the suspension.
Partial suspension
(2) In the case where the market authorization is suspended in part, a person does not contravene subsection (1) if they sell or advertise a version of the biocide other than the version described in
- (a) the notice referred to in subsection 20(1) or paragraph 21(c); or
- (b) the information provided to them under subparagraph 21(a)(i) or paragraph 21(b).
Power to suspend
20 (1) The Minister may, by written notice provided to the holder of the market authorization for a biocide, suspend the authorization in whole or in part if
- (a) the Minister has reasonable grounds to believe that the holder or an importer has, in respect of the biocide, contravened any provision of the Act or these Regulations or any order made under the Act;
- (b) the Minister has reasonable grounds to believe that the risks associated with the biocide — or, in the case of a partial suspension, the risks associated with certain conditions of use, formulants or other aspects of the biocide — outweigh the benefits associated with the biocide, taking into account any uncertainties relating to the benefits and risks; or
- (c) in the case where the market authorization was issued on the basis of an application submitted under section 25, the Minister becomes aware that the authorization to sell the foreign biocide has been revoked or suspended.
Precondition
(2) Before suspending a market authorization in whole, the Minister must consider if a partial suspension would be sufficient to address the situation giving rise to the proposed suspension.
Content of notice
(3) The notice referred to in subsection (1) must set out
- (a) the reasons for the suspension;
- (b) a statement of whether the market authorization is suspended in whole or in part and, if it is suspended in part, a description of the version of the biocide to which the suspension relates; and
- (c) the effective date of the suspension.
Serious and imminent risk
(4) The Minister may make the suspension effective immediately if the Minister has reasonable grounds to believe that the suspension is necessary to prevent a serious and imminent risk of injury to human health.
Notice to third parties
21 If the market authorization for a biocide is suspended,
- (a) the holder of the authorization must
- (i) without delay after receiving the notice referred to in subsection 20(1), provide the information that is set out in the notice under paragraphs 20(3)(b) and (c) — and, in the case of a partial suspension, the lot numbers of the version of the biocide to which the suspension relates — to any importer of the biocide and any person, other than a user, to whom the holder has sold the biocide or the affected version, as the case may be, and
- (ii) in the case where the holder is required to provide information to importers or other persons under subparagraph (i), provide to the Minister, within two business days after receiving the notice referred to in subsection 20(1) or within any longer period specified by the Minister, written confirmation that the information has been provided to those persons;
- (b) any person that receives the information referred to in subparagraph (a)(i) from the holder or from any person that has sold them the biocide must provide the information without delay to any person, other than a user, to whom the recipient of the information has sold the biocide or the affected version, as the case may be; and
- (c) the Minister must publish a notice of the suspension that sets out
- (i) a summary of the reasons for the suspension, and
- (ii) the information referred to in paragraphs 20(3)(b) and (c).
Reinstatement
22 (1) The Minister must reinstate a suspended market authorization if the situation that gave rise to the suspension has been corrected or if the reasons for the suspension were unfounded.
Notice of reinstatement
(2) If a market authorization is reinstated, the Minister must publish a notice to that effect.
Revocation
Automatic revocation
23 (1) A market authorization, or a part of a market authorization, that is suspended for a reason set out in paragraph 20(1)(a) or (c) is revoked if
- (a) in the case of a suspension under paragraph 20(1)(a), the situation that gave rise to the suspension is not corrected within the six-month period that begins on the effective date of the suspension; and
- (b) in the case of a suspension under paragraph 20(1)(c), the situation that gave rise to the foreign revocation or suspension is not corrected within the six-month period that begins on the effective date of the suspension of the market authorization.
Power to revoke
(2) The Minister may revoke a market authorization, or a part of a market authorization, that is suspended for the reason set out in paragraph 20(1)(b) if the Minister has reasonable grounds to believe that the holder has failed, within the six-month period that begins on the effective date of the suspension, to establish that the benefits associated with the biocide outweigh the risks associated with it — or, in the case of a partial suspension, the risks that led to the suspension — taking into account any uncertainties relating to the benefits and risks.
Clarification
(3) For greater certainty, the Minister is not authorized under subsection (2) to revoke a larger part of the market authorization than the part that is suspended.
Public notice
24 If a market authorization for a biocide is revoked under section 23, the Minister must
- (a) publish a notice of revocation that sets out
- (i) a summary of the reasons for the revocation,
- (ii) a statement of whether the authorization is revoked in whole or in part and, if it is revoked in part, a description of the version of the biocide to which the revocation relates, and
- (iii) the effective date of the revocation; and
- (b) amend the information that is published under subsection 11(2) accordingly.
Use of Foreign Decisions
Application
25 (1) Subject to subsection (2), a person may submit to the Minister an application for a market authorization for a biocide that is based on a comparison to a biocide that is authorized for sale
- (a) by a foreign regulatory authority named in the List of Foreign Regulatory Authorities; and
- (b) under a statute or other legislative instrument that is set out in that list in connection with the regulatory authority.
Exceptions
(2) An application cannot be submitted under subsection (1) if
- (a) the foreign regulatory authority has authorized the sale of the foreign biocide on the basis of a type of application that is set out in the List of Foreign Regulatory Authorities in connection with the regulatory authority; or
- (b) the biocide for which the market authorization is sought is a biocide referred to in subsection 61(1) or 63(1).
Content of application
(3) The application must contain
- (a) the information referred to in paragraphs 9(1)(a) to (g), (j), (k) and (m) to (o) in respect of the biocide for which the market authorization is sought;
- (b) information relating to the size and type of the package of the biocide and a statement of all of the properties and qualities of the packaging material;
- (c) subject to subsection (4), an attestation by an individual who has authority to bind the applicant confirming that the applicant possesses or has immediate access to the information referred to in paragraphs 9(1)(h) and (l) that was submitted to the foreign regulatory authority to obtain the authorization to sell the foreign biocide;
- (d) information that demonstrates that
- (i) the biocide contains, in comparison to the foreign biocide, the same active ingredients in the same quantities in a given quantity of biocide,
- (ii) the formulants contained in the biocide are among the formulants that are permitted to be contained in the foreign biocide under the foreign authorization to sell,
- (iii) the quantity of each formulant contained in a given quantity of the biocide is the same as the quantity of that formulant that is permitted to be contained in the same given quantity of the foreign biocide, and
- (iv) the biocide has the same conditions of use as the foreign biocide;
- (e) an attestation by an individual who has authority to bind the applicant confirming that the biocide will be manufactured in accordance with the master formula for the foreign biocide;
- (f) a list of the tests and studies that have been submitted to the foreign regulatory authority in connection with the authorization to sell the foreign biocide;
- (g) information that demonstrates that the sale of the foreign biocide is authorized by the foreign regulatory authority;
- (h) any terms and conditions that have been imposed by the foreign regulatory authority on the authorization to sell the foreign biocide; and
- (i) the text approved by the foreign regulatory authority for every label to be used in connection with the foreign biocide.
Exception — packaging
(4) The attestation referred to in paragraph (3)(c) does not need to confirm that the applicant possesses or has immediate access to any information relating to packaging that was submitted to the foreign regulatory authority.
Additional information and material
(5) The Minister may request the applicant to provide any additional information or any material, including samples, that is necessary to enable the Minister to determine whether the market authorization must be issued.
Follow-up obligations
26 (1) The holder of a market authorization for a biocide that is issued on the basis of an application submitted under section 25 must
- (a) subject to subsection (2), possess or have immediate access to the information referred to in paragraphs 9(1)(h) and (l) that was submitted to the foreign regulatory authority named in the market authorization to obtain the authorization to sell the foreign biocide;
- (b) provide the Minister, on request, with any information referred to in paragraph (a) within 48 hours after the request is made or any longer period specified by the Minister;
- (c) inform the Minister without delay if they become aware that
- (i) the foreign biocide has been recalled, or
- (ii) the foreign regulatory authority has revoked or suspended the authorization to sell the foreign biocide; and
- (d) provide the Minister, without delay after becoming aware that the foreign regulatory authority has required a change in respect of the foreign biocide, with a written description of the change and the reasons for it, if they are known.
Exception
(2) The holder is not required to possess or have immediate access to information relating to packaging that was submitted to the foreign regulatory authority.
Labelling and Packaging
Definitions
27 The following definitions apply in this section and in sections 28 to 36.
- designated container
- means a disposable metal container that is designed to release pressurized contents by the use of a manually operated valve that forms an integral part of the container. (contenant désigné)
- household biocide
- means a biocide that is intended to be distributed only to the general public for personal use. (biocide domestique)
- immediate container
- means the package that is in direct contact with a biocide. (contenant immédiat)
- inner label
- means a label that is on or attached to the immediate container of a biocide. (étiquette intérieure)
- outer label
- means a label that is on or attached to the outside of a package, other than the immediate container, of a biocide. (étiquette extérieure)
- Pressurized Containers Labelling Document
- means the document entitled Labelling Requirements for Pressurized Containers Containing Biocides, dated December 18, 2021, published by the Government of Canada. (Document sur l’étiquetage des contenants sous pression)
- principal display panel
- means the part of the label that
- (a) is on or attached to all or part of the surface of the package that is displayed or visible under normal conditions of sale or use; or
- (b) if the package does not have a surface described in paragraph (a), is on or attached to any part of the package except the bottom. (espace principal)
Requirement for label
28 A biocide must have an inner label and, if the immediate container of the biocide is packaged in another package, an outer label.
Bilingual labelling
29 The information that is required to be shown on a label of a biocide must be clearly and prominently displayed in both English and French.
Principal display panel
30 (1) The following information must be shown on the principal display panel of the inner label of a biocide and, if there is an outer label, on the principal display panel of the outer label:
- (a) the brand name of the biocide;
- (b) at least one of the intended uses or purposes of the biocide;
- (c) the settings in which the biocide is intended to be used;
- (d) the biocide’s identification number, preceded by the designation “DIN” in capital letters;
- (e) the net quantity of the biocide in the package;
- (f) the statements “Keep out of reach of children.” and “Tenir hors de la portée des enfants.”; and
- (g) if the biocide is sterile, the words “sterile” and “stérile”.
Limit
(2) If the biocide has more than one brand name, only one of those names may be shown on the label that is displayed or visible under normal conditions of sale.
Additional information
31 (1) The following information must be shown on the inner label of a biocide — and, if there is an outer label, on the outer label — but is not required to be shown on the principal display panel:
- (a) the biocide’s conditions of use, other than
- (i) the conditions of use that are required to be shown on the principal display panel under paragraphs 30(1)(b) and (c), and
- (ii) the directions for disposal;
- (b) the quantity of each active ingredient that is contained in a given quantity of the biocide;
- (c) the lot number of the biocide;
- (d) the expiry date of the biocide, unless the biocide is a household biocide with a shelf life of more than one year and the package contains a quantity of the biocide that is reasonably expected to be used within one year after it is sold;
- (e) the name and contact information of the holder of the market authorization for the biocide; and
- (f) the physical form of the biocide, unless it is obvious.
Contact information — accessibility
(2) For the purposes of paragraph (1)(e), the contact information must provide a means for persons in Canada to contact the holder of the market authorization without cost.
Small packages
32 In the case where the immediate container of a biocide is not large enough to accommodate an inner label that complies with sections 30 and 31, those sections do not apply in respect of the inner label if
- (a) the immediate container is packaged in another package and the outer label complies with the labelling requirements of these Regulations; and
- (b) the inner label shows
- (i) the brand name of the biocide,
- (ii) the quantity of each active ingredient that is contained in a given quantity of the biocide,
- (iii) the net quantity of the biocide that is contained in the immediate container,
- (iv) the settings in which the biocide is intended to be used, and
- (v) the information referred to in paragraphs 31(1)(c) to (f).
Pressurized containers — explosion hazard
33 (1) Subject to subsections 35(1) and (3), the following information must be shown, in accordance with sections 1 to 4 of the Pressurized Containers Labelling Document, on the principal display panel of the inner label of a biocide that is packaged in a designated container and, if there is an outer label, on the principal display panel of the outer label:
- (a) the hazard symbol set out in column 2 of item 4 of Schedule 2 to the Consumer Chemicals and Containers Regulations, 2001;
- (b) the signal words “CAUTION” and “ATTENTION”; and
- (c) the primary hazard statements “CONTAINER MAY EXPLODE IF HEATED.” and “CE CONTENANT PEUT EXPLOSER S’IL EST CHAUFFÉ.”.
Additional statements
(2) Subject to subsection 35(2), the following statements must be shown, in accordance with section 5 of the Pressurized Containers Labelling Document, on one panel of the inner label of a biocide that is packaged in a designated container and, if there is an outer label, on one panel of the outer label:
“Contents under pressure. Do not place in hot water or near radiators, stoves or other sources of heat. Do not puncture or incinerate container or store at temperatures over 50°C.”
“Contenu sous pression. Ne pas mettre dans l’eau chaude ni près des radiateurs, poêles ou autres sources de chaleur. Ne pas percer le contenant, ni le jeter au feu, ni le conserver à des températures dépassant 50 °C.”.
Pressurized containers — flammability
34 (1) Subject to subsections 35(1) and (3), where a biocide that is packaged in a designated container has a flame projection or flashback, the following information must be shown, in accordance with sections 1 to 4 of the Pressurized Containers Labelling Document, on the principal display panel of the inner label and, if there is an outer label, on the principal display panel of the outer label:
- (a) if the length of the flame projection is less than 15 cm and there is no flashback,
- (i) the hazard symbol set out in item 1 of the annex to the Pressurized Containers Labelling Document,
- (ii) the signal words “CAUTION” and “ATTENTION”, and
- (iii) the primary hazard statements “FLAMMABLE” and “INFLAMMABLE”;
- (b) if the length of the flame projection is 15 cm or more but less than 45 cm and there is no flashback,
- (i) the hazard symbol set out in item 2 of the annex to the Pressurized Containers Labelling Document,
- (ii) the signal words “WARNING” and “AVERTISSEMENT”, and
- (iii) the primary hazard statements “FLAMMABLE” and “INFLAMMABLE”; and
- (c) if the length of the flame projection is 45 cm or more or there is a flashback,
- (i) the hazard symbol set out in column 2 of item 3 of Schedule 2 to the Consumer Chemicals and Containers Regulations, 2001,
- (ii) the signal word “DANGER”, and
- (iii) the primary hazard statements “EXTREMELY FLAMMABLE” and “EXTRÊMEMENT INFLAMMABLE”.
Additional statements
(2) Subject to subsection 35(2), the following statements must be shown, in accordance with section 5 of the Pressurized Containers Labelling Document, on one panel of the inner label of a biocide described in subsection (1) and, if there is an outer label, on one panel of the outer label:
“Do not use in presence of open flame or spark.”
“Ne pas utiliser en présence d’une flamme nue ou d’étincelles.”.
Definitions
(3) The following definitions apply in this section.
- flame projection
- means the flame resulting from the ignition of a biocide discharged from a designated container when tested in accordance with official method DO-30, Determination of Flame Projection, dated October 15, 1981, published by the Government of Canada. (projection de la flamme)
- flashback
- means the part of a flame projection that extends from the point of ignition back to the designated container when tested in accordance with official method DO-30, Determination of Flame Projection, dated October 15, 1981, published by the Government of Canada. (retour de flamme)
Exceptions — small quantities
35 (1) The primary hazard statements set out in paragraph 33(1)(c) and subparagraphs 34(1)(a)(iii), (b)(iii) and (c)(iii) may be omitted from the inner label of a biocide if the net quantity shown on the inner label does not exceed 60 mL or 60 g.
Inner label — 120 mL or g
(2) The statements set out in subsections 33(2) and 34(2) may be omitted from the inner label of a biocide if the net quantity shown on the inner label does not exceed 120 mL or 120 g.
Outer label — 120 mL or g
(3) The information referred to in subsections 33(1) and 34(1) may be omitted from the outer label of a biocide if the net quantity shown on the inner label does not exceed 120 mL or 120 g.
Packaging standards
36 The immediate container of a biocide must be constructed to
- (a) contain the biocide safely under normal conditions of storage, display and distribution;
- (b) permit the user to withdraw the biocide in a safe manner and to close the container in a manner that will contain the biocide safely under normal storage conditions; and
- (c) minimize changes to, including degradation of, its contents.
Information Requirements
Notable and Serious Incidents
Definitions
37 The following definitions apply in this section and sections 38 and 39.
- notable incident
- means
- (a) a response to a biocide that adversely affects human health; or
- (b) a failure in the effectiveness of a biocide that, in respect of human health,
- (i) could have resulted in in-patient hospitalization, prolongation of existing hospitalization, congenital malformation or chronic or significant disability or incapacity, or
- (ii) could have been life-threatening or resulted in death. (événement notable)
- serious incident
- means a response to or a failure in the effectiveness of a biocide that, in respect of human health,
- (a) results in in-patient hospitalization, prolongation of existing hospitalization, congenital malformation or chronic or significant disability or incapacity; or
- (b) is life-threatening or results in death. (événement grave)
- serious unexpected incident
- means a serious incident that is not identified in nature, severity or frequency in the risk information that is set out on the Canadian label of the biocide. (événement grave et imprévu)
Serious incident reporting
38 The holder of the market authorization for a biocide must provide the Minister with the information in their control about the following serious incidents within 15 days after becoming aware of the incident:
- (a) any serious incident involving the biocide that occurs in Canada; and
- (b) any serious unexpected incident involving the biocide that occurs outside Canada.
Information relating to incidents
39 (1) The holder of the market authorization for a biocide must compile and review, in a manner that ensures the effective and timely detection of significant safety issues associated with the biocide, information that they become aware of relating to notable incidents and serious incidents involving the biocide that occur in or outside Canada, including information about measures that have been taken outside Canada to address such issues.
Significant safety issue
(2) If, in reviewing the information, the holder concludes that there is a significant safety issue related to the risks or benefits associated with the biocide, they must notify the Minister in writing without delay.
Retention period
(3) Subject to subsection (4), the holder must retain the compiled information for at least 25 years after becoming aware of it, even if they cease to hold the market authorization.
New holder
(4) If, during the 25-year period, another person becomes the holder of the market authorization, the former holder must transfer the compiled information to the new holder and the new holder must retain it for the remainder of the period.
Request for information
(5) If the Minister becomes aware of new information relating to the risks or benefits associated with the biocide, the Minister may request in writing that the holder provide the Minister with any of the compiled information that is relevant to those risks or benefits.
Time limit
(6) The Minister must specify a period within which the information is to be provided that is reasonable in the circumstances.
Obligation to provide information
(7) The holder must provide the Minister with the requested information within the specified period.
Assessments, Tests and Studies
Issue-related report
40 (1) If the Minister becomes aware of an issue that could have significant implications for the benefits or risks associated with a biocide, the Minister may, for the purpose of assessing those implications, request in writing that the holder of the market authorization for the biocide provide the Minister with a report that contains
- (a) a concise, critical analysis of the issue specified in the request;
- (b) the information on which the analysis is based; and
- (c) any additional information specified by the Minister that is accessible to the holder.
Time limit
(2) The Minister must specify a period within which the report is to be provided that is reasonable in the circumstances.
Obligation to provide report
(3) The holder must provide the report within the specified period.
Assessment order — condition
41 Before making an order under section 21.31 of the Act in respect of a biocide, the Minister must have reasonable grounds to believe that the benefits or risks associated with the biocide are significantly different than they were when the market authorization for the biocide was issued.
Order requiring tests and studies — conditions
42 Before making an order under section 21.32 of the Act in respect of a biocide, the Minister must
- (a) have reasonable grounds to believe that there are significant uncertainties relating to the benefits or risks associated with the biocide; and
- (b) take into account
- (i) whether the activities that the holder of the market authorization will be ordered to undertake are feasible, and
- (ii) whether there are less burdensome ways of obtaining the information to be provided under the order.
Hospital Reporting
Serious adverse drug reactions — biocides
43 (1) For the purposes of section 21.8 of the Act as it relates to biocides, hospitals are prescribed health care institutions that must provide the following information about a serious adverse drug reaction, in writing, within 30 days after the day on which the reaction is first documented within the hospital:
- (a) the name and civic address of the hospital and the name and contact information of a representative of the hospital;
- (b) the brand name and identification number of the biocide involved in the reaction, if known;
- (c) the age and sex of the patient who experienced the reaction;
- (d) a description of the reaction;
- (e) the date on which the patient was exposed to the biocide, if known;
- (f) the date on which the reaction first occurred, if known;
- (g) the date on which the reaction was first documented within the hospital;
- (h) if applicable, the date on which the patient’s health was restored to its state prior to the reaction;
- (i) any medical condition of the patient that directly relates to the reaction; and
- (j) any other factor that may have contributed to the reaction.
Exemption
(2) Despite section 21.8 of the Act, a hospital is required to provide the information referred to in subsection (1) only if the hospital is able to establish the biocide’s brand name or identification number.
Definition of hospital
(3) In this section, hospital means a facility
- (a) that is licensed, approved or designated as a hospital by a province in accordance with the laws of the province to provide care or treatment to individuals suffering from any form of disease or illness; or
- (b) that is operated by the Government of Canada and that provides health services to in-patients.
Clarifications
(4) For greater certainty,
- (a) in this section, serious adverse drug reaction has the same meaning as in subsection 1(2); and
- (b) nothing in this section affects the application of section C.01.020.1 of the Food and Drug Regulations.
Sales-related Information
Information — first sale
44 The holder of the market authorization for a biocide must provide the Minister with the following information in writing within 30 days after the day on which the biocide, as packaged and labelled for sale to consumers, is first sold in Canada:
- (a) the biocide’s identification number;
- (b) the date of the first sale;
- (c) the names and contact information of the persons that are packaging or labelling the biocide, excluding individuals who are employees or agents of such persons, and the civic addresses of the locations where the biocide is packaged or labelled; and
- (d) a copy of all labels that are being used in connection with the biocide.
Annual notification
45 The holder of the market authorization for a biocide must, before October 1 of each year and in the manner specified by the Minister, inform the Minister whether they are selling the biocide in Canada or have sold it in Canada in the 12 months preceding that date.
Permanent discontinuance of sale
46 The holder of the market authorization for a biocide — or, if the market authorization has been revoked, the former holder — must provide the Minister with the following information in writing within 30 days after the day on which they permanently discontinue the sale of the biocide in Canada:
- (a) the biocide’s identification number;
- (b) the date on which they discontinued the sale of the biocide; and
- (c) the latest expiry date of the biocide that they have sold and the applicable lot number.
Recalls
Recall system
47 (1) Any person that has sold a biocide must maintain a system of control, including records, that permits the complete and rapid recall of the biocide from the persons to whom they have sold it, other than consumers that purchased the biocide at the retail level.
Retention period
(2) The person must retain each record
- (a) in the case where an expiry date is shown on the label of the biocide to which the record relates, for a period that ends no earlier than the last day of the month that is shown as the expiry date; and
- (b) in any other case, for at least six years after the day on which the person sold the biocide to which the record relates.
Voluntary recall
48 (1) If any of the following persons decides to recall a biocide that is the subject of a market authorization without being ordered to do so by the Minister, the person must, within 24 hours after making the decision, provide the Minister with the information set out in subsection (2), in writing:
- (a) the holder of the market authorization;
- (b) an importer of the biocide; or
- (c) a person that manufactures the biocide and sells it.
Information to be provided
(2) The information to be provided is as follows:
- (a) the biocide’s brand names;
- (b) the biocide’s identification number;
- (c) the names and contact information of the persons that manufactured the biocide, excluding individuals who are employees or agents of such persons, and, in the case of a recall by the holder, the names and contact information of any importers;
- (d) the name and contact information of the individual who is responsible for the recall;
- (e) the lot numbers, dates of manufacture and expiry dates of the biocide that is being recalled;
- (f) the quantity of the biocide that was manufactured in Canada;
- (g) the quantity of the biocide that was imported;
- (h) the quantity of the biocide that the person sold to persons in Canada;
- (i) the period during which the person sold the biocide in Canada;
- (j) the quantity of the biocide that the person exported from Canada, as well as the quantity exported, by country;
- (k) the quantity of the biocide that is in Canada and that remains in the possession or control of the person;
- (l) the names of the persons in Canada, other than consumers that purchased the biocide at the retail level, to whom the biocide was sold by the person and the quantity sold to each of the named persons;
- (m) the classes of persons from whom the biocide is being recalled;
- (n) the expected dates for the start and completion of the recall;
- (o) the reason for the recall and the date on which, and the manner in which, the situation that prompted the recall was discovered;
- (p) an assessment of the risk of injury to human health posed by the biocide that is being recalled, including because of a failure of its effectiveness; and
- (q) a description of any other measures that the person is taking in respect of the recall.
Communications
(3) The person must
- (a) before starting the recall, provide the Minister with a copy of the communications that they intend to use in connection with starting the recall; and
- (b) after starting the recall, provide the Minister, on request and within the time specified by the Minister, with a copy of any additional communications that they use, or intend to use, in connection with the recall.
Information requirements — 72 hours
(4) The person must, within 72 hours after making the decision to recall the biocide, provide the Minister with the following information in writing:
- (a) the strategy for conducting the recall, including the time and manner in which the Minister will be informed of the progress of the recall; and
- (b) a description of the measures that are intended to be taken to prevent a recurrence of the situation that prompted the recall.
Information requirements — after recall
(5) The person must, within 30 days after completing the recall, provide the Minister with the following information in writing:
- (a) the results of the recall; and
- (b) a description of the measures that have been or will be taken to prevent a recurrence of the situation that prompted the recall.
Recall ordered by Minister
49 (1) A person that is ordered by the Minister to recall a biocide under section 21.3 of the Act must provide the Minister with the following information in the time and manner specified by the Minister:
- (a) the name, title and contact information of an individual from whom the Minister may obtain additional information concerning the recall;
- (b) if known, the names and contact information of the persons that manufactured the biocide that is being recalled, excluding individuals who are employees or agents of such persons, and the names and contact information of any importers;
- (c) the total quantity of the biocide that has been sold by the person at the retail level to consumers in Canada;
- (d) if the person has sold the biocide to persons in Canada other than consumers referred to in paragraph (c),
- (i) the names of each of those persons and the quantity of the biocide that has been sold to each of them, and
- (ii) the period during which the biocide was sold to those persons;
- (e) the quantity of the biocide that the person has exported from Canada, as well as the quantity exported, by country;
- (f) the quantity of the biocide that is in Canada and that remains in the possession or control of the person;
- (g) the strategy for conducting the recall;
- (h) any other information that the Minister has reasonable grounds to believe is necessary to mitigate the risk of injury to health; and
- (i) if the person is in a position to prevent a recurrence of the situation that prompted the recall, a description of the measures that they intend to take to prevent a recurrence.
Change to information — representative
(2) The person must notify the Minister without delay of any change to the information referred to in paragraph (1)(a).
Communications
(3) The person must
- (a) before starting the recall, provide the Minister with a copy of the communications that they intend to use in connection with starting the recall; and
- (b) after starting the recall, provide the Minister, on request and within the time specified by the Minister, with a copy of any additional communications that they use, or intend to use, in connection with the recall.
Notification of start and completion
(4) The person must notify the Minister in writing, within 24 hours, of the start and completion of the recall.
Information requirements — after recall
(5) The person must, within 30 days after completing the recall, provide the Minister with the following information in writing:
- (a) the results of the recall; and
- (b) if the person is in a position to prevent a recurrence of the situation that prompted the recall, a description of the measures that they have taken or will take to prevent a recurrence.
Quality Control
Prohibition — failure to follow master formula
50 (1) It is prohibited for the holder of the market authorization for a biocide or the importer of a biocide to sell the biocide unless the biocide has been manufactured, packaged, labelled and stored in accordance with the master formula for the biocide.
Prohibition — manufacturer, packager or labeller
(2) It is prohibited for a person that manufactures, packages or labels a biocide that is the subject of a market authorization to sell the biocide if they have failed to manufacture, package or label it, as the case may be, in accordance with the master formula for the biocide or have failed to store it in accordance with the master formula.
Records — quality control
51 (1) The holder of the market authorization for a biocide and the importer of a biocide must maintain records that demonstrate that each lot or batch of the biocide that they sell has been manufactured, packaged, labelled and stored in accordance with the master formula for the biocide.
Records — manufacturer, packager or labeller
(2) Any person that manufactures, packages or labels a biocide that is the subject of a market authorization must, for each lot or batch of the biocide that they sell, maintain records demonstrating that they have manufactured, packaged or labelled, as the case may be, the lot or batch in accordance with the master formula for the biocide and have stored it in accordance with the master formula.
Retention period
(3) The holder, importer or person must — even if they cease to hold the market authorization or no longer import or sell the biocide, as the case may be — retain each record for at least one year after the last day of the month in which the shelf life of the biocide expires or, if that day cannot be determined, for at least six years after the day on which they sold the biocide.
Requirement to provide information
(4) Anyone that is required to retain a record under subsection (3) must provide the information in the record to the Minister, on request, within five days after the request is made or, if the Minister has reasonable grounds to believe that there is a serious and imminent risk of injury to human health, within any shorter period specified by the Minister.
Prohibition — sterile biocides
52 It is prohibited to manufacture or package a biocide that is intended to be sterile unless the manufacturing or packaging is carried out
- (a) in a separate and enclosed area;
- (b) under the supervision of an individual who is trained in microbiology; and
- (c) using a method that is scientifically proven to ensure the sterility of the biocide.
Investigation of complaints
53 (1) If any of the following persons receives a complaint in respect of the quality of a biocide that is the subject of a market authorization, the person must investigate it and, if necessary, take corrective action:
- (a) the holder of the market authorization;
- (b) an importer of the biocide; or
- (c) a person that manufactures, packages or labels the biocide and sells it.
Records
(2) The person must maintain a record of each complaint and investigation and any corrective action taken.
Retention period
(3) The person must — even if they cease to hold the market authorization or no longer import or sell the biocide, as the case may be — retain each record for at least one year after the latest of
- (a) the last day of the month in which the shelf life of the biocide to which the complaint relates expires, if that day can be determined,
- (b) if no corrective action is taken, the day on which the investigation is completed, and
- (c) if corrective action is taken, the day on which the action is completed.
Requirement to provide information
(4) Anyone that is required to retain a record under subsection (3) must provide the information in the record to the Minister, on request, within five days after the request is made or, if the Minister has reasonable grounds to believe that there is a serious and imminent risk of injury to human health, within any shorter period specified by the Minister.
[54 to 60 reserved]
Transitional Provisions
Temporary exemption — pest control products
61 (1) A biocide is exempt from the application of these Regulations, other than sections 9 and 10, if, immediately before the first anniversary of the day on which these Regulations are registered, the biocide is a pest control product, as defined in subsection 2(1) of the Pest Control Products Act, that is registered under that Act.
End of exemption
(2) The exemption ceases to apply on the earliest of
- (a) the day on which the pest control product ceases to be registered under the Pest Control Products Act,
- (b) the day on which the Minister issues or refuses to issue a market authorization for the biocide,
- (c) the day on which an application for a market authorization for the biocide is withdrawn, and
- (d) the fifth anniversary of the day on which these Regulations are registered.
Exception — pest control products
62 The information referred to in paragraphs 9(1)(h), (i) and (l) may be omitted from an application for a market authorization for a biocide that is exempt from the application of these Regulations under subsection 61(1) if the conditions of use of the biocide that are set out in the application are the same as the relevant conditions, other than conditions relating to labelling, specified for the pest control product under paragraph 8(1)(a) of the Pest Control Products Act.
Temporary exemption — drug identification number
63 (1) A biocide is exempt from the application of these Regulations, other than sections 9 and 10, if, immediately before the first anniversary of the day on which these Regulations are registered, the biocide is a drug for which
- (a) a drug identification number has been assigned under subsection C.01.014.2(1) of the Food and Drug Regulations; and
- (b) the drug identification number has not been cancelled.
End of exemption
(2) The exemption ceases to apply on the earliest of
- (a) the day on which the drug identification number is cancelled under section C.01.014.6 of the Food and Drug Regulations,
- (b) the day on which the Minister issues or refuses to issue a market authorization for the biocide,
- (c) the day on which an application for a market authorization for the biocide is withdrawn, and
- (d) the fifth anniversary of the day on which these Regulations are registered.
Exception — drug identification number
64 The information referred to in paragraphs 9(1)(h), (i), (l) and (n) may be omitted from an application for a market authorization for a biocide that is exempt from the application of these Regulations under subsection 63(1) if the conditions of use of the biocide that are set out in the application are the same as the conditions of use that are authorized under the Food and Drug Regulations.
Amendments to these Regulations
65 Subsection 9(1) of these Regulations is amended by adding “and” at the end of paragraph (n) and by repealing paragraphs (p) and (q).
66 Subsection 25(2) of these Regulations is replaced by the following:
Exception
(2) An application cannot be submitted under subsection (1) if the foreign regulatory authority has authorized the sale of the foreign biocide on the basis of a type of application that is set out in the List of Foreign Regulatory Authorities in connection with the regulatory authority.
Consequential Amendments
Food and Drugs Act
Food and Drug Regulations
67 Subsection C.01.004.02(6) of the Food and Drug Regulations footnote 42 is amended by adding “and” at the end of paragraph (a), by striking out “and” at the end of paragraph (b) and by repealing paragraph (c).
68 Paragraphs C.01.014.1(2)(c) and (d) of the Regulations are replaced by the following:
- (c) the recommended route of administration;
69 Subsection C.01.040.2(5) of the Regulations is repealed.
70 The definition antimicrobial agent in subsection C.01A.001(1) of the Regulations is repealed.
71 Subsection C.01A.002(1) of the Regulations is amended by adding “and” at the end of paragraph (c), by striking out “and” at the end of paragraph (d) and by repealing paragraph (e).
72 Section C.02.002.1 of the Regulations is repealed.
Natural Health Products Regulations
| Item | Substances |
|---|---|
| 9 | A drug that is sold or represented for use in destroying or inactivating micro-organisms, or in reducing or controlling their number, on a non-living and non-liquid surface |
Pest Control Products Act
Pest Control Products Regulations
74 (1) Paragraphs 3(1)(d) and (e) of the Pest Control Products Regulations footnote 44 are replaced by the following:
- (d) a pest control product that is a drug, as defined in section 2 of the Food and Drugs Act, that is used to destroy or inactivate micro-organisms, or to reduce or control their number, on a non-living and non-liquid surface, other than a pest control product that is a biocide, as defined in subsection 1(1) of the Biocides Regulations, to which the exemption in subsection 61(1) of those Regulations applies;
(2) Paragraph 3(1)(d) of the Regulations is replaced by the following:
- (d) a pest control product that is a drug, as defined in section 2 of the Food and Drugs Act, that is used to destroy or inactivate micro-organisms, or to reduce or control their number, on a non-living and non-liquid surface;
(3) Subsection 3(2) of the Regulations is replaced by the following:
Exemption for named uses only
(2) A pest control product that is exempt under paragraph (1)(c) or (d) is exempt only in respect of any use described in that paragraph.
Coming into Force
First anniversary of day of registration
75 (1) Subject to subsection (2), these Regulations come into force on the first anniversary of the day on which they are registered.
Fifth anniversary of day of registration
(2) Sections 65 to 72 and subsection 74(2) come into force on the fifth anniversary of the day on which these Regulations are registered.
Terms of use and Privacy notice
Terms of use
It is your responsibility to ensure that the comments you provide do not:
- contain personal information
- contain protected or classified information of the Government of Canada
- express or incite discrimination on the basis of race, sex, religion, sexual orientation or against any other group protected under the Canadian Human Rights Act or the Canadian Charter of Rights and Freedoms
- contain hateful, defamatory, or obscene language
- contain threatening, violent, intimidating or harassing language
- contain language contrary to any federal, provincial or territorial laws of Canada
- constitute impersonation, advertising or spam
- encourage or incite any criminal activity
- contain a language other than English or French
- otherwise violate this notice
The federal institution managing the proposed regulatory change retains the right to review and remove personal information, hate speech, or other information deemed inappropriate for public posting as listed above.
Confidential Business Information should only be posted in the specific Confidential Business Information text box. In general, Confidential Business Information includes information that (i) is not publicly available, (ii) is treated in a confidential manner by the person to whose business the information relates, and (iii) has actual or potential economic value to the person or their competitors because it is not publicly available and whose disclosure would result in financial loss to the person or a material gain to their competitors. Comments that you provide in the Confidential Business Information section that satisfy this description will not be made publicly available. The federal institution managing the proposed regulatory change retains the right to post the comment publicly if it is not deemed to be Confidential Business Information.
Your comments will be posted on the Canada Gazette website for public review. However, you have the right to submit your comments anonymously. If you choose to remain anonymous, your comments will be made public and attributed to an anonymous individual. No other information about you will be made publicly available.
Comments will remain posted on the Canada Gazette website for at least 10 years.
Please note that public email is not secure, if the attachment you wish to send contains sensitive information, please contact the departmental email to discuss ways in which you can transmit sensitive information.
Privacy notice
The information you provide is collected under the authority of the Financial Administration Act, the Department of Public Works and Government Services Act, the Canada–United States–Mexico Agreement Implementation Act,and applicable regulators’ enabling statutes for the purpose of collecting comments related to the proposed regulatory changes. Your comments and documents are collected for the purpose of increasing transparency in the regulatory process and making Government more accessible to Canadians.
Personal information submitted is collected, used, disclosed, retained, and protected from unauthorized persons and/or agencies pursuant to the provisions of the Privacy Act and the Privacy Regulations. Individual names that are submitted will not be posted online but will be kept for contact if needed. The names of organizations that submit comments will be posted online.
Submitted information, including personal information, will be accessible to Public Services and Procurement Canada, who is responsible for the Canada Gazette webpage, and the federal institution managing the proposed regulatory change.
You have the right of access to and correction of your personal information. To seek access or correction of your personal information, contact the Access to Information and Privacy (ATIP) Office of the federal institution managing the proposed regulatory change.
You have the right to file a complaint to the Privacy Commission of Canada regarding any federal institution’s handling of your personal information.
The personal information provided is included in Personal Information Bank PSU 938 Outreach Activities. Individuals requesting access to their personal information under the Privacy Act should submit their request to the appropriate regulator with sufficient information for that federal institution to retrieve their personal information. For individuals who choose to submit comments anonymously, requests for their information may not be reasonably retrievable by the government institution.